Use of Artificial Intelligence Embryo Selection Based on Static Images to Predict First-Trimester Pregnancy Loss
Chavez-Badiola et al., Reproductive BioMedicine Online (RMBO), 2024
Table Of ContentsAbstract
Research questionCan an artificial intelligence embryo selection assistant predict the incidence of first-trimester spontaneous abortion using static images of IVF embryos?
DesignIn a blind, retrospective study, a cohort of 172 blastocysts from IVF cases with single embryo transfer and a positive biochemical pregnancy test was ranked retrospectively by the artificial intelligence morphometric algorithm ERICA. Making use of static embryo images from a light microscope, each blastocyst was assigned to one of four possible groups (optimal, good, fair or poor), and linear regression was used to correlate the results with the presence or absence of a normal fetal heart beat as an indicator of ongoing pregnancy or spontaneous abortion, respectively. Additional analyses included modelling for recipient age and chromosomal status established by preimplantation genetic testing for aneuploidy (PGT-A).
ResultsEmbryos classified as optimal/good had a lower incidence of spontaneous abortion (16.1%) compared with embryos classified as fair/poor (25%; OR = 0.46, P = 0.005). The incidence of spontaneous abortion in chromosomally normal embryos (determined by PGT-A) was 13.3% for optimal/good embryos and 20.0% for fair/poor embryos, although the difference was not significant (P = 0.531). There was a significant association between embryo rank and recipient age (P = 0.018), in that the incidence of spontaneous abortion was unexpectedly lower in older recipients (21.3% for age ≤35 years, 17.9% for age 36–38 years, 16.4% for age ≥39 years; OR = 0.354, P = 0.0181). Overall, these results support correlation between risk of spontaneous abortion and embryo rank as determined by artificial intelligence; classification accuracy was calculated to be 67.4%.
ConclusionsThis preliminary study suggests that artificial intelligence (ERICA), which was designed as a ranking system to assist with embryo transfer decisions and ploidy prediction, may also be useful to provide information for couples on the risk of spontaneous abortion. Future work will include a larger sample size and karyotyping of miscarried pregnancy tissue.
KEY WORDSArtificial intelligence • Embryo morphology • Miscarriage • Assisted conception • Embryo ranking
Introduction
Spontaneous abortion, otherwise known as early miscarriage or pregnancy loss in the first trimester, is a common complication of pregnancy (Kolte et al., 2015). The miscarriage rate is reported as 10–20% (with the lower figure pertaining to late pregnancy recognition) among naturally conceived pregnancies (Cohain et al., 2017). It has been suggested that pregnancies conceived through IVF have a slightly higher risk of miscarriage, although numbers vary (Bashiri et al., 2018). Spontaneous abortion can be a devastating experience for patients, leading to a range of stressful emotions and grief, with many couples giving up on further treatment before they become parents (Domar et al., 2018; Rooney et al., 2022). The management of spontaneous abortion also increases the time until the next opportunity for pregnancy, thus prolonging the infertility journey (Munné et al., 2019). The risk of miscarriage increases with maternal age, with the most common cause being chromosomal abnormality, principally aneuploidy (Cimadomo et al., 2018; Gruhn et al., 2019). With the exception of donor egg cycles, oocyte age is closely related to recipient age, with most patients receiving their own eggs from fresh or frozen cycles. Currently, the most common intervention for poor-prognosis patients with a high likelihood of spontaneous abortion is preimplantation genetic testing for aneuploidy (PGT-A). PGT-A is perhaps the most controversial procedure in assisted reproductive technology (ART) because of its invasive nature (embryo biopsy is required), its expense, and mixed messages from randomized controlled trials regarding its ability to improve (cumulative) pregnancy rates. As such, the Human Fertilization and Embryology Authority (HFEA), the UK regulator, has variously assigned PGT-A a red/amber light, several red lights and (currently) a green light for reducing the likelihood of miscarriage for most patients with infertility (https://www.hfea.gov.uk/treatments/treatment-add-ons/pre-implantation-genetic-testing-for-aneuploidy-pgt-a/). The HFEA system for rating 'add-ons' is no longer referred to as 'traffic lights', but uses a five-category rating system.
Nowadays, artificial intelligence applications are widely used in many areas of clinical medicine and medical research (Curchoe et al., 2020; Yu et al., 2018). ART lends itself particularly well to artificial intelligence applications, as it usually generates treatment-associated data in a variety of formats including images (from light microscope and camera), video, natural language and time. All of these parameters can potentially be used to train machine-learning algorithms to predict treatment success at different stages. Examples of these include artificial-intelligence-enhanced embryo selection (Bori et al., 2021; Chavez-Badiola et al., 2020; Tran et al., 2019; VerMilyea et al., 2020), sperm selection (Zhang et al., 2021), semen analysis (Hicks et al., 2019; Mahdavi et al., 2011) and many others (Curchoe et al., 2020a,b; Kragh et al., 2021; Riegler et al., 2021). Artificial intelligence is attractive because it facilitates the interrogation of large amounts of data, far more than the human eye can see or the mind is able to process, in a very rapid time frame. It thereby creates the possibility of identifying patterns and correlations amongst features not previously recognized as being important (Bori et al., 2020; Gardner et al., 2000).
ERICA (IVF 2.0 Ltd, UK) is an artificial intelligence system initially trained to anticipate the ploidy potential of blastocysts based on an assessment of either single static images or single images it extracts from time-lapse videos and metadata (Chavez-Badiola et al., 2020). ERICA first extracts texture patterns from the static images (light microscope and standard camera). This comprises a pre-processing step in which the images are adjusted to standardize the pixel to micrometre ratio, and each image is then convoluted with kernel filters, and segmented into four regions of interest (i.e. background, zona pellucida, trophectoderm and inner cell mass). A feature extractor designed to quantify predictors of embryo viability (e.g. size, shape of inner cell mass, and total energy and entropy measurements) is used to generate a feature vector. Next, ERICA ranks embryos based on the identification and scoring of blastocysts using extracted image-based features, and combining them with the metadata for each embryo using a binary classification model generated by a deep neural network. A total of 94 features have been extracted successfully by ERICA using these deep neural networks and artificial vision. The resulting model classification and confidence defines a score for each blastocyst in a given cycle, and ranks them according to their prognosis. In a previous study (Chavez-Badiola et al., 2020), these features were introduced into the training software, achieving accuracy of 0.70 in the validation set, and a positive predictive value for euploidy (as determined by PGT-A) of 0.79 in the test set. The ERICA algorithm assigns each embryo a value between 0 and 1, based on its putative anticipated euploidy potential, categorizing it as either optimal, good, fair or poor. The purpose is to assist embryologists in ranking the order in which blastocysts from a given cohort are to be transferred (Chavez-Badiola et al., 2020).
Based on the known association between aneuploidy and spontaneous abortion, the objective of this research was to perform an initial feasibility study to determine whether embryo ploidy ranking using ERICA could potentially be used to predict the occurrence of spontaneous abortion. Specifically, this study tested the hypothesis that events occurring between implantation (defined as a positive biochemical pregnancy result) and clinical pregnancy [defined as the detection of a fetal heart beat (FHB)] correlated with the optimal/good/fair/poor prediction using ERICA. This study also investigated whether the artificial intelligence assessment was more predictive for different recipient age groups, and whether correlations improved for embryos known to be euploid following PGT-A screening. If these hypotheses were correct, such information could be beneficial when counselling couples (or individuals) regarding their embryo quality.
Methods
A retrospective cohort of patients undergoing ART in two Mexican fertility clinics was included in this study. Only single embryo transfer cases that proceeded to biochemical pregnancy were included in the analysis.
Ethical approval, data collection, definitions and inclusion criteriaInstitutional Review Board approval was granted for this project (CONBIOETICA 09-CEI-00120170131, approval date 14 June 2021).
A database of 525 embryo transfers performed between August 2019 and May 2021 at two IVF clinics in Mexico (New Hope Fertility Centers, Guadalajara and Mexico City), for which images had been uploaded into the ERICA web application prior to their transfer (erica.embryoranking.com), was used in this study. A positive biochemical pregnancy was defined as quantitative beta-human chorionic gonadotrophin (β-HCG) ≥20 IU/l 7 days post-transfer. After accounting for 15 cases which were lost to follow-up, the biochemical pregnancy rate per embryo transfer was 39.6% (n = 248/510). FHB was defined as either present or absent, based on week 7–8 transvaginal ultrasonography (TVS) (3–4 weeks after biochemical pregnancy test). Spontaneous abortion was defined as either: an initial positive pregnancy test (β-HCG ≥20 IU/l) followed by a subsequent fall in β-HCG concentration; transvaginal bleeding equal to or heavier than a menstrual period after excluding the possibility of ectopic pregnancy; the absence of a gestational sac at TVS performed 2–3 weeks after first biochemical pregnancy test; or a pregnancy which failed to develop an FHB identifiable with TVS by 7–8 weeks. An ongoing pregnancy was defined as the presence of an identifiable FHB during TVS performed 2–4 weeks following embryo transfer (pregnancy week 8–10).
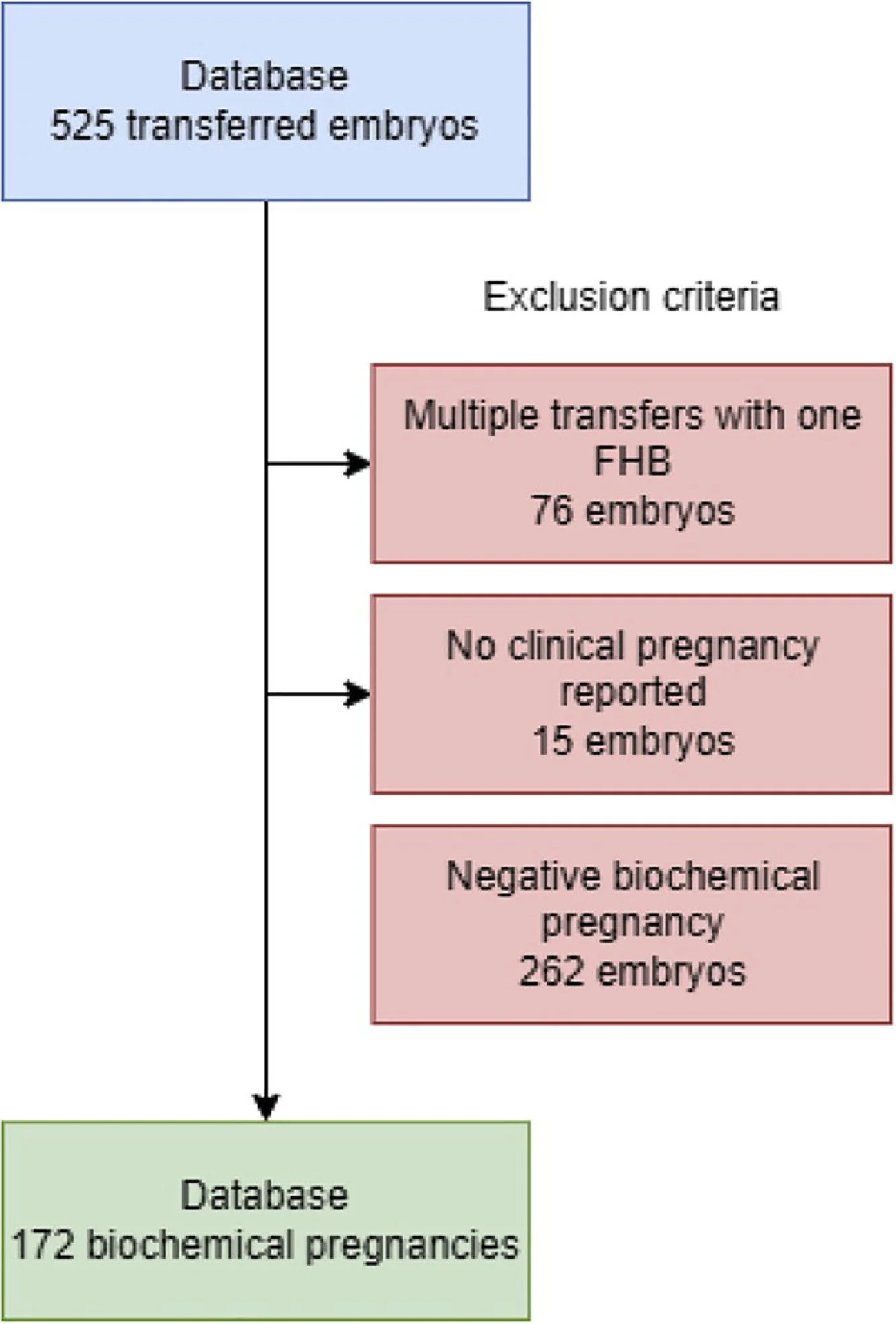
All single embryo transfer cycles with a positive biochemical pregnancy test, known FHB status (present or absent), and registered in the ERICA web application were included in the analysis. Pregnancies with a confirmed spontaneous abortion following a positive biochemical test were included even if TVS was not performed. After the exclusion criteria were applied, 172 single embryo transfers were suitable for analysis (Figure 1).
Figure 1 Number of embryo transfer cycles performed in the study period (August 2019–May 2021) and detailed inclusion process. FHB, fetal heart beat.
Ovarian stimulation and embryo cultureThe stimulation, fertilization and culture processes followed standard protocols. In brief, following stimulation and oocyte retrieval (Zhang et al., 2016), oocytes were placed in human tubal fluid medium (Life Global, EUA) for 2 h before cumulus stripping. Metaphase II oocytes were inseminated by intracytoplasmic sperm injection, and then cultured in a continuous single culture complete medium with human serum albumin (FUJIFILM IrvineScientific, Inc., EUA), covered with Life-Guard high-viscosity oil (Life Global) for incubation for 5–6 days (CO2 8.5%, O2 5%) (Tri-gas; Astec, Japan). The culture medium was renewed after the third day of incubation.
Embryo transferFresh embryo transfers were performed 5 days after oocyte retrieval. Luteal support, started on the night of oocyte retrieval, was 400 mg vaginal progesterone (Geslutin; Asofarma, USA) every 12 h until the pregnancy test, and then until week 8 if pregnant. For frozen embryo transfers, endometrial preparation was started with 4 mg oral oestradiol (Primogyn; Schering AG, Colombia) from day 3 of menstrual flow, and supplemented with 400 mg vaginal progesterone (Geslutin; Asofarma) every 12 h starting on cycle day 14–16; both continued until the pregnancy test. If the pregnancy test was positive, progesterone was continued until week 8, following the same schedule. Blastocysts were thawed 2–4 h before embryo transfer on day 6 from the beginning of luteal support. Vitrification and warming were performed using the Cryotop method and vitrification/thawing kits (Kitazato, Japan), as described elsewhere (Kuwayama et al., 2005, 2007).
Embryo imaging and artificial-intelligence-assisted rankingPictures from blastocyst-stage embryos were taken before any interventions were performed (i.e. biopsy, vitrification or transfer). Single static images were uploaded into the ERICA web application (erica.embryoranking.com) for evaluation. Embryos were photographed with total magnification of either 200X or 400X using standard cameras (ZILOS-tk Laser; Hamilton Thorne, USA) installed on inverted microscopes (IX71 and IX73; Olympis, Japan) equipped with Hoffmann modulation contrast.
Embryos were assigned an output value ranging from 0 to 1 based on their calculated individual ploidy potential. This score was used to rank all embryos within a given cohort. A technical description of ERICA is given elsewhere (Chavez-Badiola et al., 2020). Based on the output of ERICA, embryos were defined arbitrarily as: optimal, score ≥0.70; good, score ≥0.50 and <0.70; fair, score ≥0.30 and <0.50; or poor, score < 0.30
Statistical analysisBased on the hypothesis that embryos with the lowest scores will have higher spontaneous abortion rates, the ERICA categories were compared for their potential link to outcomes, defined as the presence or absence of an FHB following an initial positive pregnancy test (biochemical pregnancy). The proportion of spontaneous abortions within each category was calculated as the total number of biochemical pregnancies with no FHB, divided by the total number of biochemical pregnancies. Data are presented as percentages with their calculated 95% CI. Due to the low sample size in some comparisons, embryos classified as optimal or good were pooled, as were embryos classified as fair or poor.
Statistical analysis was completed using SPSS v.28 (IBM, USA), and made use of a generalized linear regression model using logit link functions for binary outcomes. The model tested for the presence of any interaction between embryo ranking and female age, as the increased risk of aneuploidy is positively correlated with age (Hassold and Hunt, 2009). Age brackets of ≤35, 36–38 and ≥39 years were chosen arbitrarily; however, it should be noted that, due to the use of donor eggs for some patients and incomplete records of the time between egg collection and transfer, it was not possible to make accurate calculations for oocyte age. Thus, recipient age alone was used in this study. Finally, a subset of embryos from this database (n = 50) had a known euploid diagnosis as detected by high-resolution next-generation sequencing PGT-A screening; the effect of this factor was also evaluated.
Results
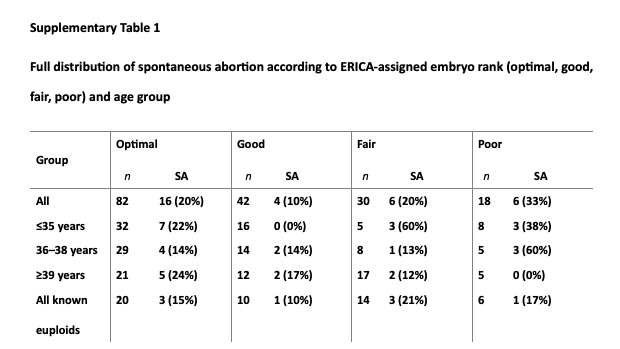
The demographics and characteristics of the biochemical pregnancies included in this study are described in Table 1. A full breakdown of the entire embryo database is presented in Supplementary Table 1 (see online supplementary material).
Table 1
Demographic information
Data are n (%) unless otherwise indicated.
a All spontaneous abortions before 7–8 weeks, including those with no identifiable fetal heart beat.
b. For clarity, ‘maternal age’ refers to recipient age in all cycles (including donor cycles).
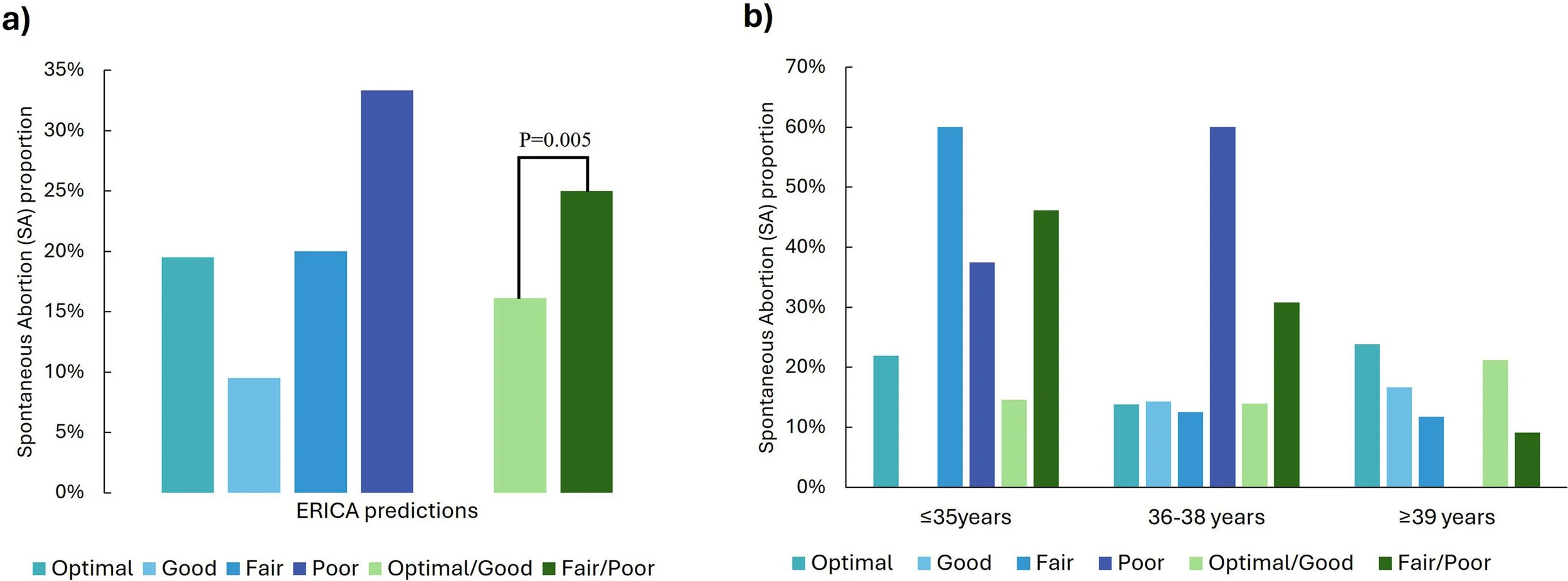
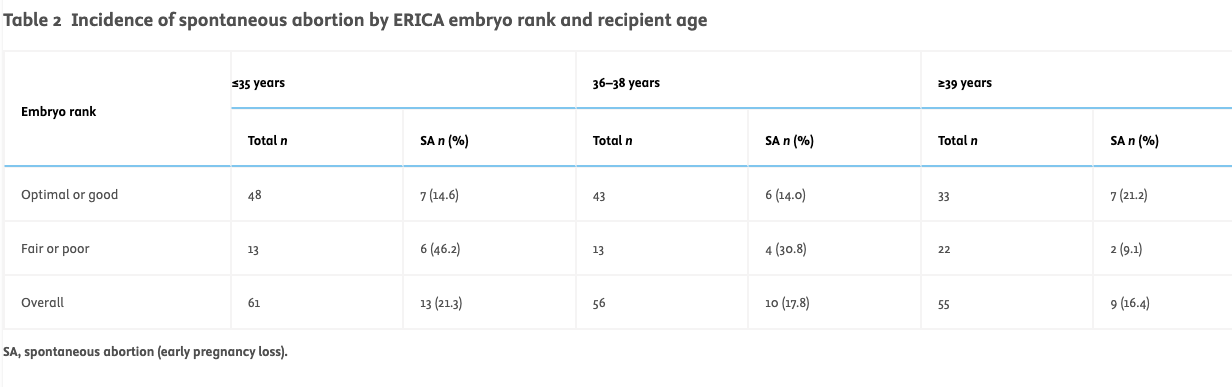
Relationship between spontaneous abortion, embryo ranking and recipient ageWhen a generalized linear model was employed, statistical analysis identified a significant effect for embryo rank. Embryos classified by the artificial intelligence assistant as either optimal or good had an incidence of spontaneous abortion of 16.1% (95% CI 10.7–23.6%, n = 20/124), compared with 25.0% for embryos classified as either fair or poor (95% CI 14.9–38.8%, n = 12/48, OR = 0.46, P = 0.005). The incidence of spontaneous abortion appeared to decrease with recipient age (21.3% for age ≤35 years, 17.9% for age 36–38 years, 16.4% for age ≥39 years; OR = 0.354, P = 0.0181). A significant interaction was detected between embryo ranking and recipient age (P = 0.018). These findings are presented in Table 2 and Figure 2.
Figure 2 Spontaneous abortion proportions by ERICA rank: (a) overall; and (b) stratified by recipient age group (≤35, 36–38 or ≥39 years). ERICA rank: optimal, score ≥0.70; good, score ≥0.50 and <0.70; fair: score ≥0.30 and <0.50; poor:
Table 2
Incidence of spontaneous abortion by ERICA embryo rank and recipient age
SA, spontaneous abortion (early pregnancy loss).
Overall, the results support some correlation between the risk of spontaneous abortion and ERICA rank. The calculated accuracy of this prediction (the overall probability that a biochemical pregnancy was correctly predicted to progress or not progress) was 67.4% (95% CI 59.9–74.4%). The test also achieved good sensitivity (74.3%, 95% CI 66.2–81.3%), but poor specificity (37.5%, 95% CI 21.1–56.3%), suggesting that the artificial intelligence was better at assigning an embryo that was associated with an ongoing pregnancy to the optimal or good rank (sensitivity) than it was at assigning an embryo that was associated with spontaneous abortion to the fair or poor rank (specificity).
Spontaneous abortion in euploid embryos determined by PGT-ANext, a sub-analysis was performed to compare the same embryo ranks and groups for known euploid embryos (n = 50). The incidence of spontaneous abortion was 13.3% (95% CI 5.3–29.7%, n = 4/30) in euploid embryos classified as either optimal or good, and 20.0% (95% CI 8.1–41.6%, n = 4/20) in embryos classified as either fair or poor; the difference was not significant (P = 0.531).
Discussion
Spontaneous abortion is a common complication encountered during fertility treatment, with an incidence rate of 12–22% in IVF cycles (Bashiri et al., 2018; Bu et al., 2020; Yang et al., 2019), although PGT-A has been shown to reduce the risk of spontaneous abortion following the transfer of euploid embryos (Lee et al., 2019; Rubio et al., 2017). Spontaneous abortion imposes a high toll on patients, not only emotionally, financially and physically, but also by increasing the time until the next opportunity for pregnancy, which, for some patients, such as those with a severely compromised ovarian reserve, could further reduce their overall chance of conceiving at all (Bashiri et al., 2018; Cohain et al., 2017; Kolte et al., 2015).
As current ART treatments tend to generate more than one transferrable embryo, use of the embryo selection process as a means to improve time to pregnancy (usually using the subjective judgement of the embryologist) has long merited attention (Lee et al., 2019; Rubio et al., 2017). As a consequence, approaches to improve this process using more objective and accurate ways of selecting viable embryos have been proposed, and include PGT-A (Munné et al., 2019), time-lapse evaluation (Kovacs et al., 2014), visual classifications (Gardner et al., 2000) and, more recently, artificial intelligence (Dimitriadis et al., 2022). Despite the controversial nature of PGT-A in its current form, it is the closest approach to a gold standard that exists for the determination of aneuploidy potential, and hence the likelihood of a pregnancy extending past the first trimester.
Embryo quality is largely considered to be the primary factor responsible for the achievement of a positive pregnancy test, although several other factors, including endometrial receptivity, may contribute (Ashary et al., 2018; Tıras et al., 2014). Embryos with severe aneuploidy, such as those with monosomies, and chaotic embryos are likely to be lost before a positive pregnancy test is achieved. Beyond this, one could assume that many of the basic factors that might have prevented implantation have been overcome successfully, such as the implantation window and the embryo transfer procedure itself. It is at this point that aneuploidy (principally, trisomy of the autosomes and monosomy X) is relevant. Although the causes of spontaneous abortion are multifactorial, aneuploidy is widely cited as the most common reason (Hassold and Hunt, 2001; Rose et al., 2020).
The curious incidence of spontaneous abortion appearing to decrease with recipient age bears further scrutiny. Ordinarily, one would expect to see an increase in spontaneous abortion associated with older oocytes (Hassold and Hunt, 2009) and, with most recipients (83.7%) receiving their own eggs, a corresponding, although perhaps less strong, association with recipient age. Unfortunately, in this study, it was not possible to determine the age of the oocytes used, partly because the ages of the donor oocytes were not recorded accurately, and partly because records of the time between egg collection and transfer were incomplete. The apparent ‘reverse maternal age effect’ recorded here could be explained by an increase in the proportion of donor eggs employed in the older patient group(s), and/or increased attention in the treatment of older patients; however, this is not clear at the time of writing, and should be logged more accurately in future studies.
Another factor worthy of consideration is the relatively small sample size of this study (32 spontaneous abortion events). In some ways, it is surprising that such high accuracy was achieved in this study, considering that the algorithm seems to classify most embryos as good or fair. Although sample size calculations were not performed in this study, one argument (which, it is appreciated, is circular) is that significant results were seen, so the sample size must have been sufficient. On the other hand, it is appreciated that, in future studies, the magnitude of the predictive effect of ERICA will be further established. This will be performed on more advanced versions of the programme, and on newly acquired morphokinetic images and videos. These will form part of newly designed studies and future publications.
ERICA was developed as a ranking tool to advise embryologists about the best order in which embryos from a cohort could be transferred. This ranking was developed using PGT-A results as its ‘ground truth’, with clinical results supporting its performance (Chavez-Badiola et al., 2020). Although ERICA was not designed to predict the risk of miscarriage per se, aneuploidy is the most common cause of spontaneous abortion. As such, this study aimed to determine whether there was a correlation between ERICA classification and spontaneous abortion, and found that the ranking assessment was somewhat predictive of spontaneous abortion, as it was able to correctly classify just over two-thirds of the embryos analysed.
Nonetheless, it is acknowledged that before a truly robust statistical evaluation of the hypothesis can be made, the limited sample size in this study should be addressed. Furthermore, while it is not claimed that ERICA is a more accurate assessment of embryo ploidy than the gold standard PGT-A, it was interesting to note a potential application for this tool for the prediction of spontaneous abortion. It should also be considered that PGT-A usually requires cryostorage and subsequent frozen embryo transfer, which are expensive and invasive procedures for the embryos. Not all embryos will survive the biopsy and warming process, and some couples only have a small number of embryos per cohort.
To the best of the authors’ knowledge, this is the first study to address the association between pregnancy loss using an artificial intelligence algorithm and static images. Recently, however, Amitai et al. (2023) identified six morphodynamic features extracted from time-lapse images that had high prediction capability for spontaneous abortion. These centred around the distribution of nucleolar precursor bodies within the small pronucleus(i) predicting miscarriage at 0.68–0.69. The authors thus claim to have developed a decision-support tool for identifying embryos with a greater chance of miscarriage. With the now-increased interest in PGT-A for reducing the likelihood of miscarriage based on the newly awarded ‘green light’ by HFEA (https://www.hfea.gov.uk/treatments/treatment-add-ons/pre-implantation-genetic-testing-for-aneuploidy-pgt-a/), the use of tools to predict miscarriage, perhaps associated with aneuploidy, is becoming more topical. Whether artificial intelligence would be used instead of, or as an adjunct to, PGT-A remains to be seen. Indeed, a better understanding of the likelihood of pregnancy and miscarriage for each embryo will assist clinical staff to best manage their patients’ expectations during counselling. For future studies focusing on miscarriage, karyotyping of the pregnancy tissue would establish whether ERICA is as good as, or nearly as good as, PGT-A to predict ploidy status, and hence the likely potential for spontaneous abortion of an IVF cycle.
Appendix Supplementary Materials
For each subset, total sample size (n) is given, together with number and calculated percentage incidence of spontaneous abortion (SA) per group. Age groups determined by recipient age (years).
-
Amitai, T. ∙ Kan-Tor, Y. ∙ Or, Y. ... Embryo classification beyond pregnancy: early prediction of first trimester miscarriage using machine learning. Journal of assisted reproduction and genetics. 2023; 40:309-322
Ashary, N. ∙ Tiwari, A. ∙ Modi, D. Embryo implantation: war in times of love. Endocrinology. 2018; 159:1188-1198
Bashiri, A. ∙ Halper, K.I. ∙ Orvieto, R. Recurrent Implantation Failure-update overview on etiology, diagnosis, treatment and future directions. Reproductive Biology and Endocrinology. 2018; 16:1-18
Bori, L. ∙ Paya, E. ∙ Alegre, L. ... Novel and conventional embryo parameters as input data for artificial neural networks: an artificial intelligence model applied for prediction of the implantation potential. Fertility and Sterility. 2020; 114:1232-1241
Bori, L. ∙ Dominguez, F. ∙ Fernandez, E.I. ... An artificial intelligence model based on the proteomic profile of euploid embryos and blastocyst morphology: a preliminary study. Reproductive BioMedicine Online. 2021; 42:340-350
Bu, Z. ∙ Hu, L. ∙ Su, Y. ... Factors related to early spontaneous miscarriage during IVF/ICSI treatment: an analysis of 21,485 clinical pregnancies. Reproductive biomedicine online. 2020; 40:201-206
Chavez-Badiola, A. ∙ Flores-Saiffe-Farías, A. ∙ Mendizabal-Ruiz, G. ... Embryo Ranking Intelligent Classification Algorithm (ERICA): artificial intelligence clinical assistant predicting embryo ploidy and implantation. Reproductive BioMedicine Online. 2020; 41:585-593
Cimadomo, D. ∙ Fabozzi, G. ∙ Vaiarelli, A. ... Impact of maternal age on oocyte and embryo competence. Frontiers in endocrinology. 2018; 9:327
Cohain, J.S. ∙ Buxbaum, R.E. ∙ Mankuta, D. Spontaneous first trimester miscarriage rates per woman among parous women with 1 or more pregnancies of 24 weeks or more. BMC pregnancy and childbirth. 2017; 17:1-7
Curchoe, C.L. ∙ Malmsten, J. ∙ Bormann, C. ... Predictive modeling in reproductive medicine: where will the future of artificial intelligence research take us? Fertility and sterility. 2020; 114:934-940
Curchoe, C.L. ∙ Farias, A.F.S. ∙ Mendizabal-Ruiz, G. ... Evaluating predictive models in reproductive medicine. Fertility and sterility. 2020; 114:921-926
Dimitriadis, I. ∙ Zaninovic, N. ∙ Badiola, A.C. ... Artificial intelligence in the embryology laboratory: a review. Reproductive biomedicine online. 2022; 44:435-448
Domar, A.D. ∙ Rooney, K. ∙ Hacker, M.R. ... Burden of care is the primary reason why insured women terminate in vitro fertilization treatment. Fertility and sterility. 2018; 109:1121-1126
Gardner, D.K. ∙ Lane, M. ∙ Stevens, J. ... Blastocyst score affects implantation and pregnancy outcome: towards a single blastocyst transfer. Fertility and sterility. 2000; 73:1155-1158
Gruhn, J.R. ∙ Zielinska, A.P. ∙ Shukla, V. ... Chromosome errors in human eggs shape natural fertility over reproductive life span. Science (New York, N.Y.). 2019; 365:1466-1469
Hassold, T. ∙ Hunt, P. To err (meiotically) is human: the genesis of human aneuploidy. Nature Reviews Genetics. 2001; 2:280-291
Hassold, T. ∙ Hunt, P. Maternal age and chromosomally abnormal pregnancies: what we know and what we wish we knew. Current opinion in pediatrics. 2009; 21:703
Hicks, S.A. ∙ Andersen, J.M. ∙ Witczak, O. ... Machine learning-based analysis of sperm videos and participant data for male fertility prediction. Scientific reports. 2019; 9:16770
Kovacs, P. Embryo selection: the role of time-lapse monitoring. Reproductive Biology and Endocrinology. 2014; 12:1-11
Kolte, A.M. ∙ Bernardi, L.A. ∙ Christiansen, O.B. ... Terminology for pregnancy loss prior to viability: a consensus statement from the ESHRE early pregnancy special interest group. Human reproduction. 2015; 30:495-498
Kragh, M.F. ∙ Karstoft, H. Embryo selection with artificial intelligence: how to evaluate and compare methods? Journal of Assisted Reproduction and Genetics. 2021; 38:1675-1689
Kuwayama, M. ∙ Vajta, G. ∙ Ieda, S. ... Comparison of open and closed methods for vitrification of human embryos and the elimination of potential contamination. Reproductive biomedicine online. 2005; 11:608-614
Kuwayama, M. Highly efficient vitrification for cryopreservation of human oocytes and embryos: the Cryotop method. Theriogenology. 2007; 67:73-80
Lee, C.I. ∙ Wu, C.H. ∙ Pai, Y.P. ... Performance of preimplantation genetic testing for aneuploidy in IVF cycles for patients with advanced maternal age, repeat implantation failure, and idiopathic recurrent miscarriage. Taiwanese Journal of Obstetrics and Gynecology. 2019; 58:239-243
Mahdavi, H.S. ∙ Monadjemi, A. ∙ Vafae, A. Sperm detection in video frames of semen sample using morphology and effective ellipse detection method. Journal of medical signals and sensors. 2011; 1:206
Munné, S. ∙ Kaplan, B. ∙ Frattarelli, J.L. ..., STAR Study Group. Preimplantation genetic testing for aneuploidy versus morphology as selection criteria for single frozen-thawed embryo transfer in good-prognosis patients: a multicenter randomized clinical trial. Fertility and sterility. 2019; 112:1071-1079
Riegler, M.A. ∙ Stensen, M.H. ∙ Witczak, O. ... Artificial intelligence in the fertility clinic: status, pitfalls and possibilities. Human Reproduction. 2021; 36:2429-2442
Rooney, K.L. ∙ Domar, A.D. The relationship between stress and infertility. Dialogues in clinical neuroscience. 2022
Rose, N.C. ∙ Kaimal, A.J. ∙ Dugoff, L. ..., American College of Obstetricians and Gynecologists. Screening for fetal chromosomal abnormalities: ACOG practice bulletin, number 226. Obstetrics & Gynecology. 2020; 136:e48-e69
Rubio, C. ∙ Bellver, J. ∙ Rodrigo, L. ... In vitro fertilization with preimplantation genetic diagnosis for aneuploidies in advanced maternal age: a randomized, controlled study. Fertility and sterility. 2017; 107:1122-1129
Tıras, B. ∙ Cenksoy, P.O. Practice of embryo transfer: recommendations during and after. Thieme Medical Publishers, 2014 291-296 No. 04
Tran, D. ∙ Cooke, S. ∙ Illingworth, P.J. ... Deep learning as a predictive tool for fetal heart pregnancy following time-lapse incubation and blastocyst transfer. Human reproduction. 2019; 34:1011-1018
VerMilyea, M. ∙ Hall, J.M.M. ∙ Diakiw, S.M. ... Development of an artificial intelligence-based assessment model for prediction of embryo viability using static images captured by optical light microscopy during IVF. Human Reproduction. 2020; 35:770-784
Yang, P. ∙ Wang, Y. ∙ Wu, Z. ... Risk of miscarriage in women with endometriosis undergoing IVF fresh cycles: a retrospective cohort study. Reproductive Biology and Endocrinology. 2019; 17:1-9
Yu, K.H. ∙ Beam, A.L. ∙ Kohane, I.S. Artificial intelligence in healthcare. Nature biomedical engineering. 2018; 2:719-731
Zhang, J.J. ∙ Merhi, Z. ∙ Yang, M. ... Minimal stimulation IVF vs conventional IVF: a randomized controlled trial. American journal of obstetrics and gynecology. 2016; 214 96-e1
Zhang, Z. ∙ Dai, C. ∙ Shan, G. ... Quantitative selection of single human sperm with high DNA integrity for intracytoplasmic sperm injection. Fertility and Sterility. 2021; 116:1308-1318
Key Message
Correlation was found between the risk of spontaneous abortion and embryo rank as determined by artificial intelligence using the ERICA algorithm, with classification accuracy of 67.4%. This preliminary study suggests that ERICA may provide useful information for couples regarding their risk of spontaneous abortion.