Automated Oocyte Retrieval, Denudation, Sperm Preparation, and ICSI in the IVF Laboratory: a Proof-of-Concept Study and Report of the First Live Births
A Chavez-Badiola, Human Reproduction, 2025
Table Of ContentsIntroduction
Materials and Methods
Results
Discussion
Acknowledgements
Abstract
Study questionCan multiple automated systems sequentially perform Day 0 IVF procedures: (i) sperm preparation, (ii) cumulus–oocyte complex (COC) retrieval and oocyte denudation, and (iii) ICSI?
SUMMARY ANSWERAutomated sequential Day 0 procedures achieved fertilization in 64.3% of injected oocytes and 42.2% usable blastocyst formation in 11 cases, resulting in five healthy live births from nine patients with positive pregnancy tests.
WHAT IS KNOWN ALREADYThe Day 0 procedures—sperm preparation, oocyte handling (e.g. finding and denudation), and ICSI—rely on embryologist skill. Automation in IVF laboratories has the potential to improve reproducibility, yet its implementation remains limited. Fully automated pipetting workflows replicating embryologist activities have not yet been achieved, though partial automation of key steps such as dish preparation and ICSI has been reported.
STUDY DESIGN, SIZE, DURATIONProof-of-concept pilot study with prospective allocation of sibling oocytes to automated or manual protocols. Here, we report on Day 0 automation in 11 cases using two or three automated systems (nicknamed ‘pearls’) in sequence. Single vitrified/warmed blastocyst transfers occurred between April and October 2024. This work was part of a larger research programme evaluating automation across multiple IVF laboratory procedures from gamete handling through blastocyst transfer and specifically demonstrates sequential automation across multiple Day 0 procedures within a single workflow.
PARTICIPANTS/MATERIALS, SETTING, METHODSEleven consenting patients (three autologous, eight donor egg cycles) underwent IVF/ICSI following minimal or mild stimulation. The gametes were processed using either automated or manual protocols. The automated systems—Pearl 1 (sperm preparation), Pearl 2 (COC retrieval and oocyte denudation), and Pearl 3 (sperm selection, laser immobilization, and piezo-ICSI)—were used in various combinations. Multiple AIs were developed and deployed across these systems. This IRB-approved study was conducted at Hope IVF, Guadalajara, México.
MAIN RESULTS AND THE ROLE OF CHANCEThe automated systems achieved 64.3% fertilization (45/70) and 42.2% usable blastocyst formation per zygote (19/45), compared to 81% (47/58) and 59.6% (28/47) with manual procedures. Transfers from the automated arm resulted in five live births, three biochemical pregnancies, and one early loss at 7 weeks. The live birth rate per transfer of a single warmed blastocyst in the automated arm was 5/12 (41.7%).
LIMITATIONS, REASONS FOR CAUTIONThe small sample size prevented statistical comparison between automated and manual procedures. Some steps required operator support via direct intervention or digital control. Autonomy (defined as automated execution without human intervention) was achieved only in sperm preparation and selected ICSI tasks. A larger study using an updated system is underway.
WIDER IMPLICATIONS OF THE FINDINGSThis study demonstrates the feasibility of automating Day 0 IVF procedures, with the potential to improve standardization and reduce protocol drift, fatigue, and operator variability. Our findings support a phased integration of automation to meet growing ART demands.
STUDY FUNDING/COMPETING INTEREST(S)The study was sponsored by Conceivable Life Sciences. A.C.-B. is an employee, shareholder, and company officer at Conceivable Life Sciences and also holds shares in IVF 2.0. G.M.-R. is a stock option holder at Conceivable Life Sciences and has received consulting fees from both Conceivable Life Sciences and IVF 2.0. A.F.-S.F. is an employee and stock option holder at Conceivable Life Sciences. N.C.-B. is a stock option holder at Conceivable Life Sciences and has received consulting fees from the company, and he is also an employee, shareholder, and company officer at Embryotools. A.M. is a shareholder and company officer at Conceivable Life Sciences and holds shares in TMRW Life Sciences. M.A. is a stock option holder at Conceivable Life Sciences and has received consulting fees and travel support from the company; she has also received consulting fees from TMRW Life Sciences. G.S. is an employee and stock option holder at Conceivable Life Sciences and has received consulting fees from IVF 2.0. C.M. is an employee and stock option holder at Conceivable Life Sciences. E.H.-M. is an employee and stock option holder at Conceivable Life Sciences. R.V.-M. is an employee and stock option holder at Conceivable Life Sciences and has received consulting fees from IVF 2.0. V.M. is an employee and stock option holder at Conceivable Life Sciences. E.M. is an employee of Embryotools. A.V.A. has no conflicts of interest to declare. V.O.-H. is an employee and stock option holder at Conceivable Life Sciences and has received consulting fees from IVF 2.0. F.A.-G. is an employee and stock option holder at Conceivable Life Sciences. A.Á.L. is an employee and stock option holder at Conceivable Life Sciences. M.A. is an employee of Embryotools. Q.M.-A. is an employee of Embryotools. J.G.E.F. is an employee and stock option holder at Conceivable Life Sciences. L.M.C.O. has no conflicts of interest to declare. C.P.B. is an employee and stock option holder at Conceivable Life Sciences. D.J.S.-G. has no conflicts of interest to declare. J.C. is an employee, shareholder, stock option holder, and company officer at Conceivable Life Sciences and holds shares in IVF 2.0, Althea Science, TMRW Life Sciences, Kindbody, and Reproductive Healthcare. Furthermore, A.C.-B., G.M.-R., A.F.-S.F., A.M., A.Á.L., and J.C. declare inventorship on US patent 12,349,940 B2 protecting automated oocyte denudation. A.C.-B., G.M.-R., A.F.-S.F., C.M., R.V.-M., V.O.-H., E.H.-M., V.M., A.M., and J.C. declare inventorship on US patent 12,245,793 B2 protecting robotic handling systems for IVF laboratories. A.C.-B., G.M.-R., A.F.-S.F., C.M., G.S., V.M., J.C., and A.M. declare inventorship on US patent 12,178,475 B1 protecting automated sperm preparation. A.C.-B., G.M.-R., A.M., and J.C. declare inventorship on US patent 12,180,441 B1 protecting laboratory workflow automation. A.C.-B., G.M.-R., A.F.-S.F., A.M., C.M., R.V.-M., V.O.-H., N.C.-B., A.Á.L., and J.C. declare inventorship on US patent 12,268,418 B2 protecting automated oocyte preparation. A.C.-B., G.M.-R., R.V.-M., V.O.-H., N.C.-B., E.H.-M., A.M., J.C., and A.F.-S.F. declare inventorship on US patent 12,310,625 B2 protecting automated ICSI. A.C.-B., G.M.-R., A.F.-S.F., C.M., V.O.-H., and A.M. declare inventorship on US patent 12,253,516 B2 protecting optical systems for IVF automation. A.C.-B., G.M.-R., A.M., J.C., A.F.-S.F., C.M., R.V.-M., V.O.-H., G.S., N.C.-B., and J.G.E.F. declare inventorship on US patent 12,226,125 B2 protecting automated vitrification. A.C.-B., G.M.-R., C.M., R.V.-M., V.O.-H., N.C.-B., V.M., and J.C. declare inventorship on US patent US 12,478,405 B2 protecting automated dish-based sperm preparation. A.C.-B., G.M.-R., C.M., N.C.-B., M.A., A.M., and J.C. declare inventorship on a pending patent application protecting automated dish preparation technology. A.C.-B., G.M.-R., N.C.-B., and J.C. declare inventorship on a pending patent application protecting automated oocyte denudation. N.C.-B., J.C., A.M., E.H.-M., and A.C.-B. declare inventorship on a pending patent application protecting automated vitrification systems. A.C.-B., G.M.-R., V.O.-H., and J.C. declare inventorship on a pending patent application protecting COC detection technology.
TRIAL REGISTRATION NUMBERNCT06074835 (ClinicalTrials.gov, 4 October 2023).
KeywordsAI, assisted reproduction, automation, embryo transfer, fertilization, ICSI, infertility, IVF/ICSI outcome, pregnancy
Introduction
The human IVF laboratory is where gametes and embryos of patients undergoing ART treatments are handled. From the laboratory’s perspective, a typical IVF treatment cycle begins one day after administration of an ovulation trigger following ovarian stimulation (OS), which is one day before ultrasound-guided follicular aspiration. This day is designated as Day minus 1 in the laboratory and requires preparation of dishes, culture, and handling media for the patient. The day of follicular aspiration is designated as developmental day zero (Day 0). From this day onwards, for the following 7–8 days, the laboratory serves as a transitory environment for holding, observation, and manipulation of these gametes and subsequently, the developing embryos until cryopreservation or intrauterine transfer of clinically usable embryos.
Although other peripheral tasks are performed during Day 0, including record keeping, data collection and management, as well as daily quality control, the main gamete-handling activities of Day 0 are sperm preparation, cumulus–oocyte complex (COC) identification and isolation, oocyte denuding in preparation for ICSI, insemination of oocytes via conventional IVF or ICSI, and placement of injected or inseminated oocytes in culture. These activities are labour-intensive, and their successful completion is dependent on the skill, knowledge, and focus level of the laboratory staff. As demand for IVF and the number and complexity of procedures increase, many IVF laboratories face the challenge of handling the additional workload and efficiently managing daily tasks without overburdening the laboratory staff (Alikani et al., 2014; Veiga et al., 2022). Ensuring optimal and consistent laboratory results is also a challenge with increased caseloads. Protocol drift, fatigue, and different levels of experience can lead to lack of control, variability in results, and even critical errors (Sakkas et al., 2018; Tiegs and Scott, 2020; Forbrig et al., 2025).
One way to meet these challenges is through the incorporation of artificial intelligence (AI) and automation in the IVF laboratory (Jiang and Bormann, 2023). While full autonomy (complete, independent machine decision-making) (Yang et al., 2017) is yet to be achieved, over the past two decades, many attempts have been made at partial automation (task autonomy) of various procedures, including semen analysis (Agarwal et al., 2019), vitrification (Roy et al., 2017), ICSI (Lu et al., 2011; Costa-Borges et al., 2023), and dish preparation (Lattin et al., 2024). However, clinical translation has been limited.
We have reported the first live birth from a fully automated remote ICSI procedure (Mendizabal-Ruiz et al., 2025), demonstrating that the performance of a technically complex procedure that involves biological processes can be automated and guided digitally by human operators, without on-site presence or the need for direct human physical manipulation. Here, we present more cases from the same pilot study focusing on multiple Day 0 procedures applied in sequence through the deployment of up to three systems, which were called ‘pearls’, automating sperm preparation (Pearl 1); follicular fluid search, COC isolation, and oocyte denudation (Pearl 2); and ICSI (Pearl 3). Pearl 1 included two systems performing sperm preparation according to the classic swim-up method (Pearl 1 SU) or using an alternative dish-based preparation method termed horizontal swim-out (HSO) (Baldini et al., 2020). Operation of these systems is described, and all cases in which two or three Pearl systems were applied (a string of pearls) during Day 0 in the laboratory are presented.
Materials and Methods
Ethical approvalsThis study was approved by an institutional IRB (Comité de ética en investigación New Hope Fertility Center, CONBIOÉTICA-09-CEI-001-20170131, Approval Reference: RA-2023-01, September 2023). The study protocol was registered with Clinicaltrials.gov (NCT06074835, release date 4 October 2023). Patient recruitment began on 23 October 2023, and the study enrolled 42 patients, of which one was eliminated from the final analysis. Written consent was obtained in all cases. The 11 patients presented here, representing all cases where at least two Pearl systems were used in sequence, were treated between April and October 2024.
Definition of termsIn this proof-of-concept study, we distinguished three levels of automation, together with the concept of intervention, to describe how the Pearl systems were deployed. These definitions are used throughout the manuscript:
Automated: A system that executes tasks through programmed instructions, sensors, and actuators but operates within fixed parameters and cannot adapt beyond its programmed responses.
Digital control: All robotic actions are initiated and continuously controlled by a human operator in real-time.
Autonomous: A system capable of independently executing specific tasks (or a set of tasks) without human intervention once initiated. The human operator has discrete, rather than continuous, input.
Task autonomy: Autonomy applied at the level of a defined procedural unit within a larger IVF workflow, such as sperm selection or microtool focusing during ICSI. The procedural unit can include action, perception, and decision-making. Task autonomy is achieved when the procedural unit completes with no human intervention.
Intervention: An action taken by a human operator to modify, override, or halt an automated system’s operation, including assuming continuous control.
Pre-clinical validation testsBefore the Pearl systems were applied clinically, a comprehensive range of studies was performed to optimize protocols and evaluate procedural safety and system efficacy.
Pre-clinical validation of robotic sperm preparationHuman donor semen samples were used for validation of sperm preparation. Robotic and control preparations were compared for motility and for the presence of seminal plasma.
For the validation of the automated swim-up procedure, n = 12 raw ejaculates were processed by the Pearl 1 SU system, which drew a 0.5 ml aliquot and overlaid it with HEPES-buffered medium. The supernatant was recovered automatically after incubation for 20 min at room temperature. A similar aliquot from the same ejaculate was also prepared by manual swim-up (Millan et al., 2024).
The automated HSO method was evaluated in a further n = 3 donor sperm samples split four ways and prepared in parallel by automated HSO (Pearl 1 HSO system), conventional density gradient centrifugation, conventional swim-up, and automated swim-up (Pearl 1 SU system). HSO preparation was completed when the sperm preparation achieved 20 or 50 spermatozoa per high-power field (20× magnification) visualized in the last channel before spermatozoa reached the final droplet.
All prepared samples were assessed for concentration and progressive motility. Additionally, to test the presence of seminal plasma components in the final preparation samples, samples were serially diluted (1:1000, 1:10 000, 1:100 000) and tested in triplicate for the presence of prostate-specific antigen (PSA) by using immunochromatographic assays with a detection limit of 50 μg/ml (Seratec Semiquant, Göttingen, Germany).
Pre-clinical validation studies of hyaluronidase exposure timeFresh mouse COCs (B6/CBA F1) were used for hyaluronidase exposure studies to investigate the effect of long exposure on developmental competence. COCs were exposed to 80 IU hyaluronidase for 1, 10, 60, or 120 min, followed by pipetting and washing in culture medium. Oocytes underwent piezo-ICSI with fresh sperm and were cultured to the blastocyst stage following established procedures (Costa-Borges et al., 2025).
Sperm immobilization by laser—feasibility evaluationA preliminary set of tests aimed at establishing feasibility evaluated laser exposure times between 0.3 and 10 mS (at 1480 nm) on mouse sperm, with shots applied to either the sperm head or the middle region of the tail. Individual spermatozoa were visually evaluated for immobilization, then injected into fresh mouse oocytes by piezo-ICSI. Blastocysts from both experimental and control (mechanical immobilization) groups were transferred, and offspring were monitored for two generations (Costa-Borges et al., 2024).
Sperm immobilization by laser—mouse oocyte activation testHuman sperm were immobilized by laser using the most effective parameters established in the previous test (1.2 mS laser shot to the tail) with either one or two laser pulses, and then injected into mouse oocytes. Control groups for injection included motile sperm, mechanically immobilized sperm, and sham injections. Oocyte activation was assessed by second polar body extrusion at 3 h, with pronuclei formation and cleavage evaluated at 24 h.
Robotic ICSI evaluationThe efficacy of the Pearl 3 system was evaluated by using a hamster oocyte/human sperm model. Oocyte survival was assessed 2 h post robotic piezo-ICSI performed by digital control by a remote operator (n = 102) versus manual injection groups (n = 102) (Mendizabal-Ruiz et al., 2025).
Automated COC identification and oocyte denudationBovine COCs were extracted from abattoir ovaries and preserved in follicular fluid at 37°C. The Pearl 2 system was tested for: (i) COC identification in follicular fluid (n = 31), (ii) COC isolation and washing in handling medium (n = 30), and (iii) oocyte denudation after 24-h in vitro maturation (n = 23). Efficiency rates, procedure times, and aspiration/deposition cycle accuracy were measured.
Random polar body position during ICSI and chance of damage to the spindleRabbit oocytes were selected due to their similarity to human oocytes in cytoplasmic consistency and spindle size. Spindle position was visualized using polarized light microscopy. First polar body position was evaluated for correlation with spindle location in MII oocytes. Random injection positioning was then compared to deliberate spindle placement in the injection path, with assessment of lysis, cleavage, and blastocyst formation rates.
Patients, controlled OS, endometrial preparation, and clinical proceduresThe study inclusion criteria were indication to perform ICSI, autologous or donor oocytes, autologous or donor sperm, and presence of motile sperm after sperm preparation. The exclusion criteria were recurrent pregnancy loss, previous history of total fertilization failure, surgical sperm retrieval, severe male factor infertility, known semen liquefaction problems, and any other complications that could impact success.
Three autologous and eight oocyte donation cases are reported here. These 11 cases were selected for this report because they represent all cases in which multiple Pearl systems were applied sequentially during a single oocyte retrieval cycle. Specifically, a proportion of oocytes and/or semen from each case underwent processing through either two or three automated Pearl systems (Pearl 1—sperm preparation, Pearl 2—COC retrieval and oocyte denudation, and/or Pearl 3—ICSI), with the remaining proportion processed manually as controls. All oocytes were fresh, and semen samples were fresh except in one case where frozen donor sperm was utilized.
Prior to OS, patients underwent contraceptive pre-treatment by taking one daily tablet of combined oral hormonal contraceptive (Levonorgestrel–Ethinyl Oestradiol; Microgynon, 0.15/0.03 mg) between days 1 and 3 of their menstrual period for 14 days. For OS, recombinant FSH (Gonal-F, Merck) was administered at 150–175 IU daily, starting on cycle Days 2–3 until ∼Days 10–12. A flexible GnRH antagonist protocol (Cetrotide, Merck) was initiated when the leading follicle reached ≥14 mm diameter. Final oocyte maturation was triggered with recombinant hCG (Ovidrel 250 μg subcutaneous, Merck) when three or more follicles exceeded 18 mm. In cases with risk of ovarian hyperstimulation syndrome, defined by serum oestradiol levels above 3500 pg/ml, final maturation was alternatively triggered with GnRH agonist (as recommended by the Practice Committee of the ASRM (Practice Committee of the American Society for Reproductive Medicine, 2024) triptorelin (Gonapeptyl Daily, Ferring). Oocyte retrieval was performed 35–36 h post-trigger.
All clinically usable embryos were vitrified on Day 5 or 6 of development for warming and transfer in a subsequent cycle.
Endometrial preparation for vitrified–warmed transfer cycles started with baseline monitoring on cycle Days 2–3, including transvaginal ultrasound to confirm endometrial thickness (<7 mm) and serum hormone measurements (oestradiol <50 pg/ml, progesterone <1 ng/ml). Oestradiol supplementation included oral oestradiol valerate (Primogyn, Bayer) 2 mg twice daily for 4 days, followed by three times daily dosing. The first follow-up occurred on cycle Days 8–10, monitoring endometrial development via ultrasound along with serum oestradiol and progesterone levels (maintained at <1.0 ng/ml). A second follow-up on cycle Days 12–13 confirmed optimal endometrial development (trilaminar pattern, homogeneous appearance, 8–10 mm thickness) and adequate oestradiol levels (≥250 pg/ml). The implantation window was then initiated with a single subcutaneous dose of recombinant hCG (Ovidrel 250 μg subcutaneous, Merck), concurrent with micronized progesterone (Crinone, Merck, 400 mg every 12 h) starting at 08:00 h. Embryo transfer was scheduled 120 h after progesterone initiation.
Both follicular aspiration and embryo transfer were done under ultrasound guidance.
Culture media, incubation, fertilization and embryo assessment, vitrification, and warmingManual procedures have been described previously (Mendizabal-Ruiz et al., 2025). The automatic and manual procedures involved the same routines:
Search of the follicular fluid.
Isolation and washing of COCs in Multipurpose Handling Medium-Complete with gentamicin and 5% human serum albumin (MHM-C; Fujifilm Irvine Scientific, Santa Ana, USA).
Transfer of COCs to culture dishes containing Continuous Single Culture Medium supplemented with 5 mg/ml human serum albumin (CSCM-C; FUJIFILM Irvine Scientific, Santa Ana, USA) under mineral oil (Hypure, Kitazato, Japan).
Incubation in an atmosphere of 9% CO2 and 5% O2 at 37°C in a humidified atmosphere (SMA-30DR, Astec, Japan). CO2 level was determined by pH testing of culture media and in consideration of the altitude of the laboratory (1590 m) and consistent with usual practice at this site (Mendizabal-Ruiz et al., 2025).
Injected oocytes were checked for fertilization 16–18 h post injection. Normally fertilized oocytes, showing two pronuclei (2PN) and, where verifiable, two polar bodies, were placed in CSCM-C for incubation. Embryo development was assessed on Days 5 and 6 of development, and embryos were scored according to the Gardner scoring system (Gardner and Schoolcraft, 1999). The embryos were also assessed using an AI algorithm, ERICA (Chavez-Badiola et al., 2020; Palmer et al., 2025) prior to vitrification. The algorithm generates a qualitative (poor, fair, optimal) and quantitative (scale of 0–1 with 1 being optimal) score to assist in ranking the embryos. The scale is as follows: Poor: 0–0.29; Fair: 0.3–0.49; Good: 0.5–0.69; and Optimal: 0.7–1.
Embryos judged as clinically usable with minimum Grade C for both the inner cell mass (ICM) and the trophectoderm (TE) according to Gardner’s criteria (Gardner and Schoolcraft, 1999) were vitrified using the Kitazato cryotop method (Kitazato, Japan, https://kitazato-ivf.com/vitrification/, accessed 18 March 2025). The Kitazato protocol was also followed for warming of embryos.
Development and operation of automated sperm preparation systems (Pearl 1)Two systems were developed, tested, and used: One system executed a swim-up method (Millan et al., 2024). A second system was developed to perform a HSO method, a technique described previously (De Martin et al., 2017; Baldini et al., 2020); the automated system’s droplet and channel configuration was based on studies led by Ferrer Buitrago (Ferrer Buitrago et al., 2023) and Ruiz-Jorro (Ruiz-Jorro et al., 2023).
The systems were designed to perform automated pipetting of precise volumes of medium and semen at a controlled rate. In the swim-up method, precise layering of medium over semen was central to the technique, avoiding mixing of the two layers, the need for centrifugation, and contamination of the final sample with seminal fluid. The HSO system setup was partially automated, as the system dispensed and connected media droplets in the required L-shaped configuration (connecting droplets of media with media channels), but operator assistance was required to introduce the necessary oil overlay.
Both automated methods incorporated AI-based robotic pipetting and performed their tasks with human supervision.
All patient and sample identification and verification steps, as well as assessment of sperm concentration, count, and motility before and after sperm preparation, were handled by human operators.
The novel robotic swim-up device incorporated the following hardware:
Metal plate maintained at 37°C (continuously monitored through sensors and a digital readout).
Custom-made, 3-D printed semen cup holder installed directly on the warming plate.
Robotic ‘rocking’ mechanism with rocking movements orchestrated using proprietary code to control position, timing, and speed of the holder.
Custom-made robotic pipettor.
3D-printed cartridge for pipette tips.
Robotic tilting mechanism for the swim-up tube with tilting movements orchestrated using proprietary code to control position, timing, and speed of the tube holder.
Tube holders.
3D-printed automated ejection mechanism.
The HSO system consisted of:
A six-degrees-of-freedom robotic arm (Mechademics, USA).
A 200 μl digital pipettor (Sartorius, Germany) attached to the end effector.
An inverted microscope (Olympus IX81, Japan) with a motorized stage (Prior Scientific), and a heated stage (Tokai Hit, Japan).
Similar to the other system, semen and media vessels were held in a custom-designed, 3D-printed rack, and movements were orchestrated using proprietary code to control position, timing, and speed.
The systems for swim-up (Pearl 1 SU) and for HSO (Pearl 1 HSO) are shown in Fig. 1.
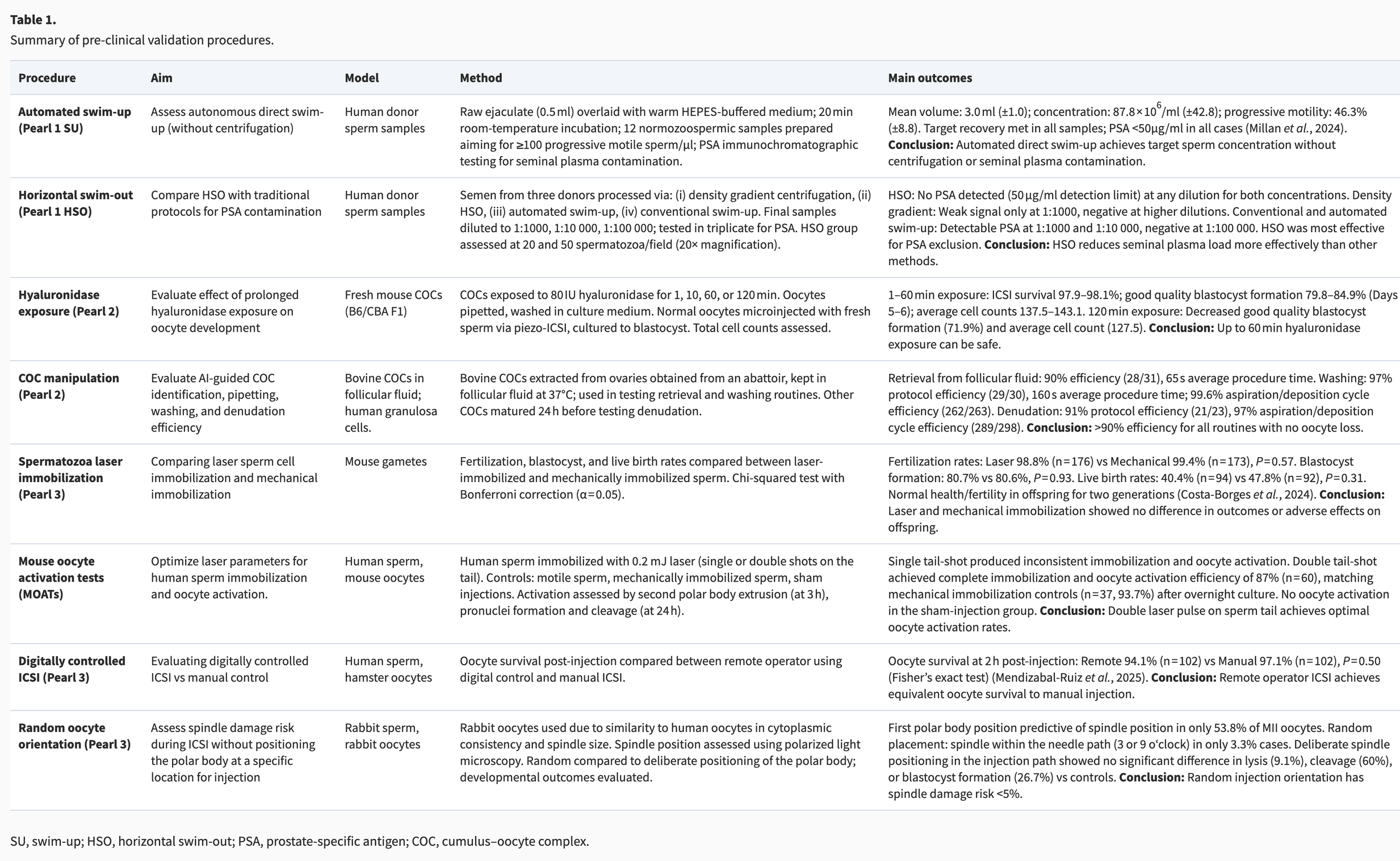
Figure 1.
Overview of automated systems for sperm preparation. (A) Pearl 1 system for swim-up (SU) featuring automated pipetting of semen and culture media, and completion of a sperm preparation procedure in a standard 15 ml conical tube. (B) Pearl 1 system for dish-based horizontal swim-out (HSO) sperm preparation featuring six-axis robotic arm, and dedicated semen cup and dish holder. (C) The HSO system autonomously produced the correct configuration of droplets to support sperm preparation by HSO. In this prototype, a human operator dispensed the oil overlay in the dish.
Swim-up method (Pearl 1 SU)The swim-up method was performed as described below and shown in Video 1.
A human operator prepared the system by first placing tubes in specified tube holders and multiple pipette tips in a tip cartridge.
Next, the operator placed the specimen cup in a cup holder on a warming plate maintained at 37°C and issued a single command to begin an autonomous sequence of steps (including a calibration routine) in the sperm preparation programme.
A robotic ‘rocking’ mechanism tilted the specimen cup to a designated degree and then brought it back to its original upright position and repeated this motion several times to facilitate liquefaction.
A robotic pipettor moved along X and Y axes to the position of the pipette tip cartridge, moved down (Z-axis) to attach a pipette tip to the pipettor, and moved back up to the zero position (uppermost position) after retrieving a tip.
Once a tip was attached, the pipettor moved to the position of the specimen cup and lowered the tip into the cup while the cup was tilted, aspirating a designated volume of semen and expelling the volume back into the specimen cup, thus mixing the specimen.
Following mixing, the pipettor lowered into the specimen cup while the cup was in the tilted position, aspirated a designated volume of semen (typically 0.5 ml), and lifted out of the specimen cup to a designated height while the robotic rocker went back to the upright position.
The pipettor then moved to a position precisely above the swim-up tube, lowered into the bottom of the tube, and expelled the semen, at a designated speed and acceleration, without creating air bubbles.
The used pipette tip was ejected into the discard bag, and a new tip was retrieved from the cartridge.
The pipettor then moved to a position directly above a tube containing MHM-Complete (Irvine Scientific, USA), lowered into the medium at a designated depth, aspirated a designated volume (typically 0.5 ml), lifted out of the tube, moved to the tube containing semen, lowered into the tube to a precise position above the semen meniscus, and ‘layered’ the medium at a designated speed and acceleration over the semen without mixing the two layers.
Once layering was completed, the pipettor moved up and out of the tube and repeated the tip ejection step to discard the used pipette tip; it remained stationary in a designated position for a designated length of time while the swim-up was underway.
After the designated time for swim-up had elapsed (typically 20 min), a new tip was retrieved, and the pipettor moved to a position directly above the swim-up tube, moved down to a specified depth of the swim-up layer and aspirated a designated volume of the cell suspension (typically 0.2 ml), then moved to a position above the final tube, lowered into the tube, and expelled the full volume of the swim-up suspension at the bottom of the tube.
In a final step, the used tip was discarded and the pipettor remained stationary in a designated position.
A human operator removed the final sperm preparation tube, and the sample was stored at room temperature for use during ICSI.
Validation of this system was performed using donated human sperm as previously described (Millan et al., 2024) and outlined in Table 1. The experiments showed that the system was able to process normozoospermic semen samples efficiently, maintaining sterility and producing a highly motile and clean sample without the need for centrifugation.
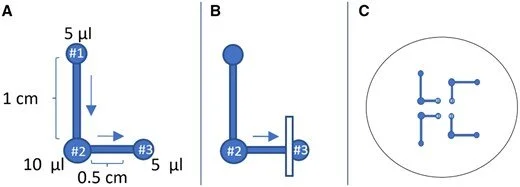
Figure 2.
Illustration of the horizontal swim-out process. (A) The HSO configuration. Following liquefaction, 2 μl aliquots of the raw ejaculate were added to #1 droplet, containing 5 μl of MHM-C. Each of these semen-containing droplets was connected via a 1 cm long horizontal channel to an intermediate droplet (#2) containing 10 μl of MHM-C. These intermediate droplets were then connected through 0.5 cm long channel at a 90-degree angle to a 5 μl 7% PVP droplet (#3). Motile sperm migrates through the connected droplets, from the raw ejaculate droplets to the last (third) drop, following the direction pointed by the arrows. (B) Illustration of channel cut position, which occurs after a predefined sperm concentration is reached. (C) Four HSO configurations in the same dish were used for each sperm preparation procedure.
This method employed an ICSI dish (Vitrolife, Sweden) with a precise arrangement of three interconnected medium droplets under light mineral oil (Kitazato) (Fig. 2A). The last droplet in the series was 7% PVP. This configuration was repeated four times in the same dish as shown in Fig. 2C.
The four configurations were automatically arranged, and channels were created between specific droplets via automated movement of a 200 μl pipette tip (devoid of medium) from one droplet to another, followed by manual overlay of 5 ml pre-warmed oil.
Semen samples were allowed to liquefy at room temperature prior to addition to the dish.
The HSO dish was pre-warmed to 37°C, and the procedure was conducted at 37°C on a heated microscope stage (Tokai Hit, Japan).
An AI system monitored the terminal droplet using a 4× objective. The HSO procedure was considered complete when at least 20 motile sperm cells were counted within a single high-power field of view in this final connection channel, a process typically requiring 20–40 min.
Upon achieving this sperm count, a 200 μl pipette tip was drawn perpendicularly across the channel to disrupt/cut them to stop the selection process and prevent diffusion of seminal plasma into the last droplet (Fig. 2B).
Operation of an automated COC retrieval and oocyte denudation system (Pearl 2)The Pearl 2 system (see Fig. 3) consisted of two Cartesian robots. The top Cartesian robot with two degrees of freedom incorporated a Full-HD camera attached to a linear motorized stage for focusing (Tango, Märzhäuser Sensotech, Germany), which could also be interchanged for a 4× magnification objective (Olympus). The top Cartesian robot also governed two micromanipulators with three degrees of freedom (Sensapex, USA) that held two pipettes connected to two 1000 µl electronic pipettors (Satorius) through flexible tubing; these pipettes could be replaced with a Narishige (Japan) tool holder for fine bore capillaries for oocyte stripping. The bottom Cartesian robot, also with two degrees of freedom, held a collimated backlight (Dinolyte, Taiwan). Dishes were held on a heated stage (Tokai Hit) supported by a custom 3D-printed template with slots for one 60 mm dish and two 35 mm dishes. The Pearl 2 system could deliver three routines: Follicular fluid search to identify COCs (R1), COC isolation and wash (R2), and oocyte denudation (R3). The operation of the Pearl 2 system is demonstrated in Video 3 and further described below:
Figure 3.
Overview of an automated system for oocyte handling and denudation (Pearl 2). (A) Enclosed system overview showing two Cartesian robots (top and bottom) separated by an intermediate heated stage for housing dishes. (B) Detailed view of the custom 3D-printed dish holder adapter, resting on the heated stage. In this configuration, the top Cartesian robot is equipped with a 4× objective and dual Narishige microtool holders to support oocyte denudation.
A human operator prepared the required dishes containing handling medium with an oil overlay and placed it on the stage with a custom-designed dish holder.
The human operator received follicular aspirate tubes from the operating room, decanted the fluid in dishes and pre-scanned for COCs using both the naked eye and stereo-microscope objectives at low magnification.
Once the operator located a COC, they placed the same dish containing the follicular fluid and COCs on the inverted microscope stage. This human intervention ensured that all COCs were identified by the AI-driven robotic system.
A command was issued on a computer by the operator, starting the automated COC identification and isolation sequence. The sequence could also be broken down to individual steps and commands.
The sequence started with movement of the stage as computer vision allowed scanning of the dish in a predetermined pattern from top to bottom; this was achieved through the integrated HD camera (the scope objective was not used).
AI identified cumulus masses containing eggs, moved the stage to place the COC at a predefined position of the visual field (close to the centre), and lowered the pipette in the dish before applying negative pressure to aspirate the COC.
The amount of fluid aspirated was precisely controlled based on the outer physical limits of the mass. Aspiration was stopped once the full mass was inside the pipette.
Then the pipette was lifted out of the dish and remained stationary while the stage was moved to bring the dish with handling medium to the field of view, the pipette with the COC was lowered into the dish, positive pressure was applied, and the COC/follicular fluid was expelled into the medium in a predetermined position.
The volume was again precisely controlled so that positive pressure was stopped once the entire mass exited the pipette.
The pipette was then lifted out of the dish and remained stationary in the air.
The stage was moved back to the follicular fluid dish, and the same actions were repeated to pick up and deposit other COCs in the wash dish.
When the automated sequence was completed, a human operator removed the dish with the remaining follicular fluid, and the same sequence was repeated until all follicular fluid tubes were searched.
Washing of COCs involved multiple aspirations and expelling in wash medium, largely replicating the motion sequence described above. To prepare oocytes for ICSI, denudation was carried out by a 3-min exposure to 80 IU/ml hyaluronidase (FujiFilm Irvine Scientific, USA) to disperse cumulus cells, followed by multiple aspirations/expulsions through a 200 and a 150 μm bore pipette to remove the corona cells. Denudation was completed between 36.5 and 38.5 h after the trigger injection under AI guidance, allowing for the automated identification, pick-up, and release of the oocytes. Complete denudation was verified and finalized manually by a human operator for every oocyte.
Operation of an automated ICSI system (Pearl 3)Development, validation, and operation of the automated ICSI system were described in an earlier report (Mendizabal-Ruiz et al., 2025).
Briefly, a standard ICSI platform was modified to include a PiezoXpert actuator (Eppendorf, Germany), a motorized stage (H117, Prior Scientific, UK), a motorized objective turret (U-D6BDREMC, Olympus), and microinjectors (Narishige), with visualization provided by Arducam IMX477 cameras. Multiple AI systems worked in parallel to manage the procedure, including the previously developed ‘SiD’ system for sperm selection (Mendizabal-Ruiz et al., 2022), alongside custom AI implementations for gamete/microtool segmentation and positioning. The system employed laser application for sperm immobilization and zona pellucida thinning, combined with piezo-assisted oolemma penetration.
Results
Average times for each step or procedure for all automated systems are provided in Supplementary Table S1.
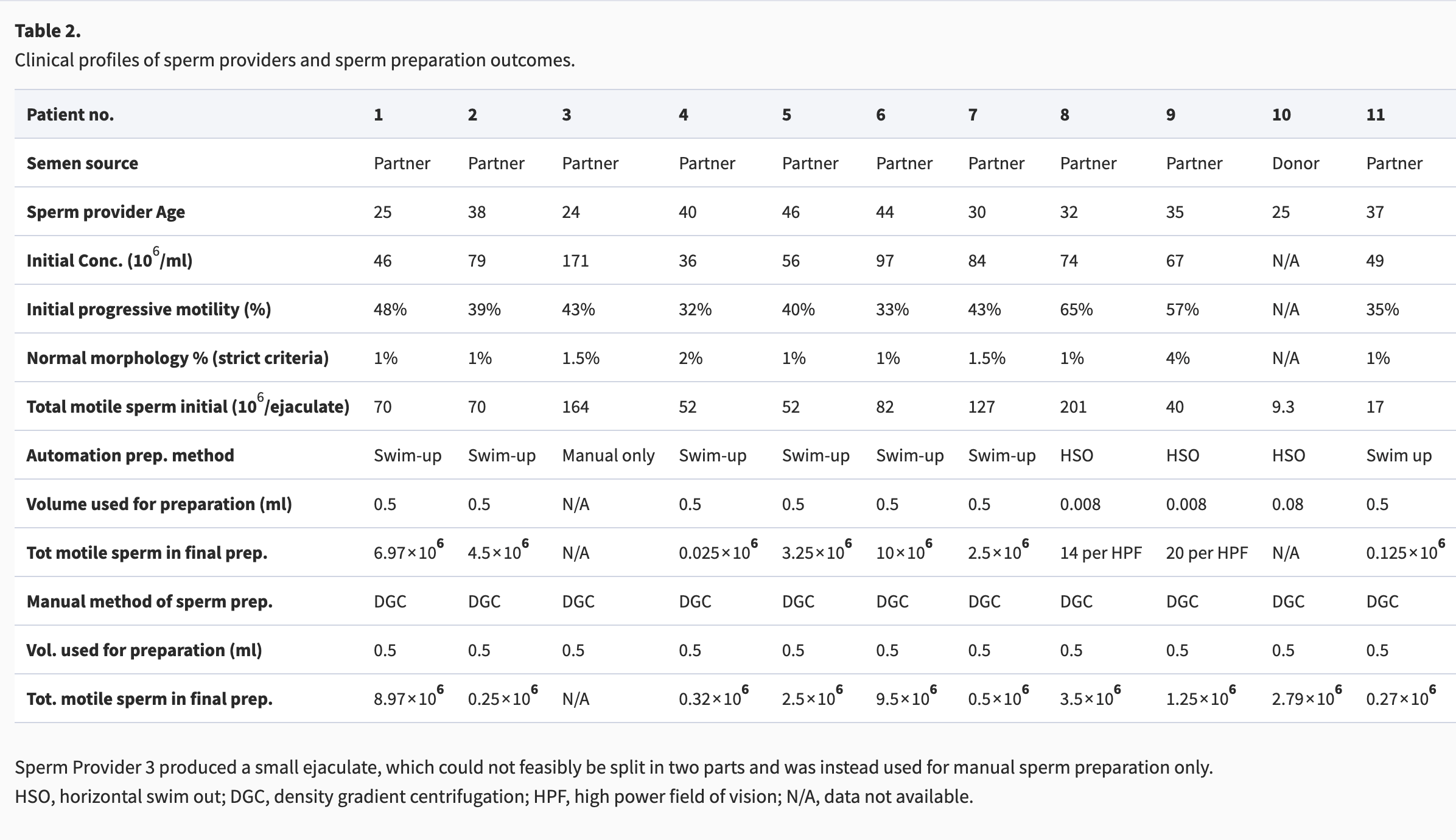
Pre-clinical validationBriefly, eight procedures across three technological ‘pearls’ were evaluated. Validation experiments and results for Pearl 1, 2, and 3 are described in Table 1.
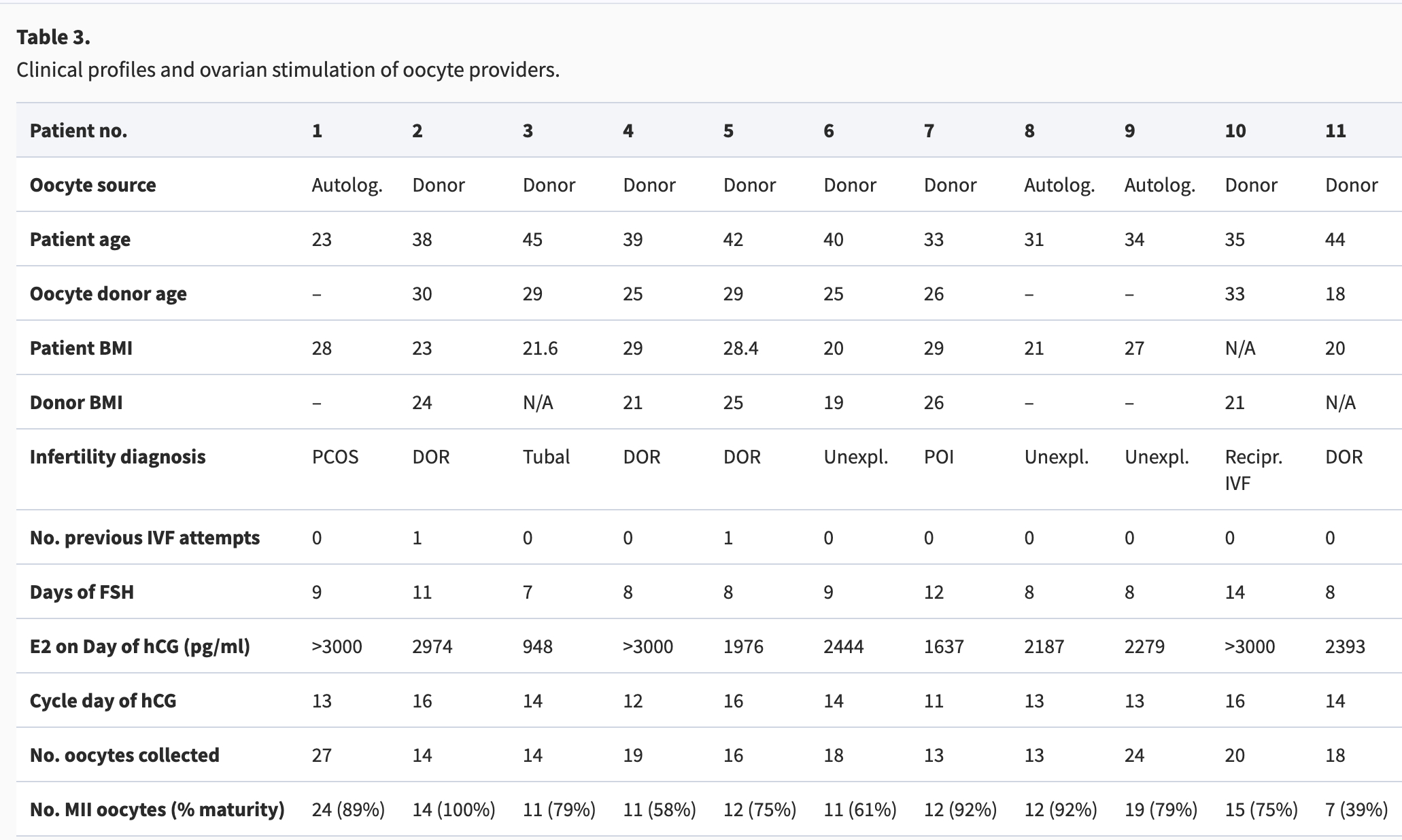
Patient characteristicsClinical profiles of sperm and oocyte providers enrolled in the proof-of-concept study are presented in Tables 2 and 3.
Table 2. Clinical profiles of sperm providers and sperm preparation outcomes.
Sperm Provider 3 produced a small ejaculate, which could not feasibly be split in two parts and was instead used for manual sperm preparation only.
HSO, horizontal swim out; DGC, density gradient centrifugation; HPF, high power field of vision; N/A, data not available.
Table 3. Clinical profiles and ovarian stimulation of oocyte providers.
Autolog, autologous; DOR, diminished ovarian reserve; E2, oestradiol; N/A, data not available; PCOS, polycystic ovary syndrome; POI, primary ovarian insufficiency; Unexpl, unexplained, idiopathic; Recipr, reciprocal.
Sperm preparationOf the 11 cases described in this study, 10 received treatment involving the use of a Pearl 1 system. All semen samples except one were split into two aliquots, one of which was processed automatically by a Pearl 1 system (either SU or HSO), and the other was processed manually. All automatically processed samples (n = 10/10) had adequate concentration and motility (total motile count) for use in ICSI. Operator intervention was required only once, during one of the n = 3 HSO procedures, due to the system adding semen to only three of the four intended locations (initial droplets) in the HSO dish (please refer to Fig. 2).
Manual preparation was performed using density gradient separation and washing. Details of the initial semen analysis and final preparation are presented in Table 2.
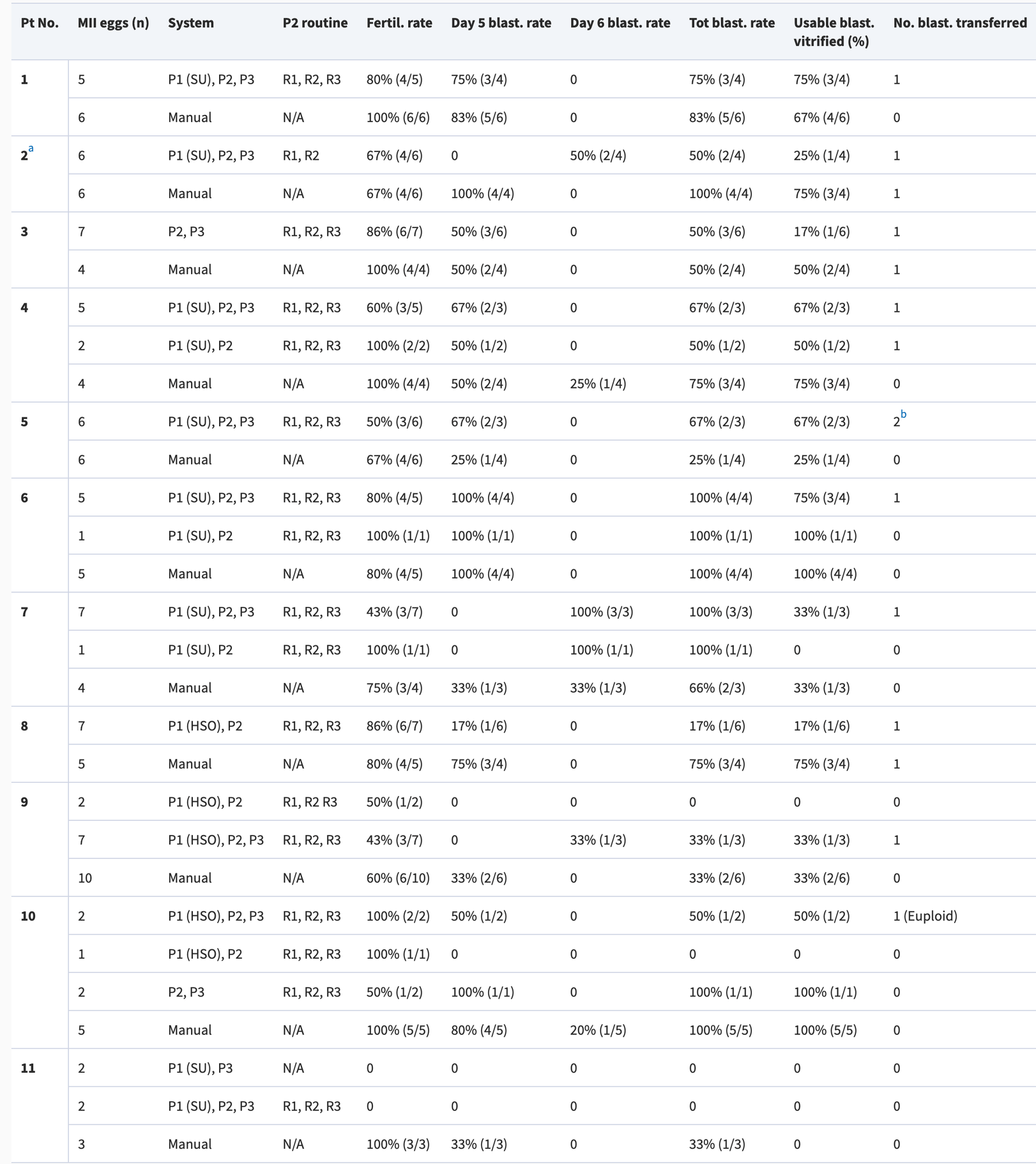
Fertilization, embryo development, and clinical outcomesThe systems utilized in each case, as well as fertilization and embryo development outcomes for automated and manual systems, are presented in Table 4. Overall, the automated systems achieved 64.3% fertilization (45/70) and 42.2% usable blastocyst formation per zygote (19/45), compared to 81% (47/58) and 59.6% (28/47) with manual procedures. Three patients had all three systems applied, while two patients had two systems applied. Six other patients had both two and three systems applied to different oocytes. The allocation of systems varied between patients due to the step-wise and careful progression of the study, where we initially tested single systems before combining multiple systems, as well as practical constraints including sample quality and system availability.
Table 4. Allocation of oocytes to automated systems or manual handling and laboratory outcomes.
Pt, patient; P1, Pearl 1, sperm preparation; SU, swim-up; HSO, horizontal swim-out; P2, cumulus–oocyte complex (COC) retrieval and oocyte denudation; R1, Routine 1, follicular fluid search; R2, Routine 2, COC isolation; R3, Routine 3, oocyte denudation; P3, Pearl 3, ICSI; N/A, not applicable.
Fertilization rate was defined as number of 2PN zygotes at 16–18 h post-insemination divided by the number of injected oocytes.
The total number of eggs reported for each patient does not necessarily match the total number of mature eggs reported in Table 3. This is because, on occasion, eggs were vitrified for future use, or were allocated to a single Pearl only, and were thus excluded from this report.
a
Two COCs from this patient were lost during manipulation with the Pearl 2 system.
b
During two separate transfers.
The Pearl 2 system was employed in all 11 cases and completed its routines without requiring direct operator intervention in 61% of tasks for follicular fluid search (R1), 67% for isolation (R2), and 71% for denudation (R3). Interventions included assuming digital control of the system. All oocytes denuded by Routine 3 were manually checked by an embryologist and further denuded to ensure the cell could be accurately segmented by the machine vision element of Pearl 3, enabling automated ICSI.
The Pearl 3 system was employed in 10 cases and completed its routines without requiring a direct operator intervention in 61% of tasks. Interventions consisted in the operator assuming digital control of the system.
An overview of task autonomy rates is given in Supplementary Table S2.
Fertilization and usable blastocyst formation in the automated and manual treatment groups were comparable as were the differences between application of two and three automated systems.
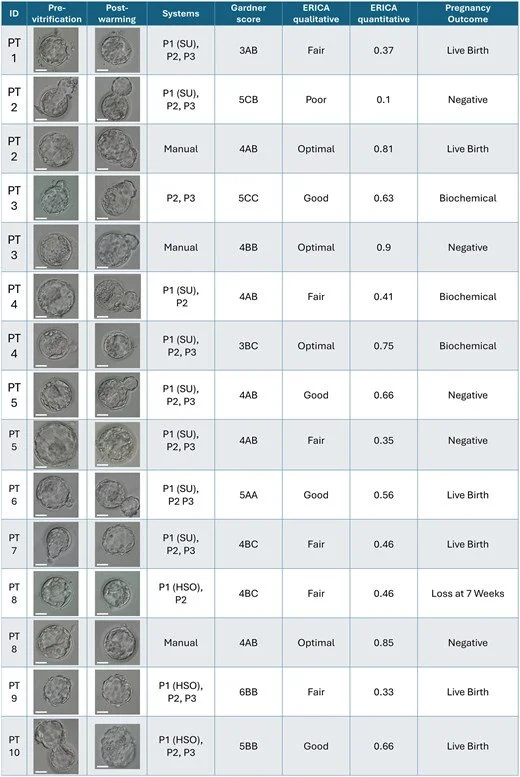
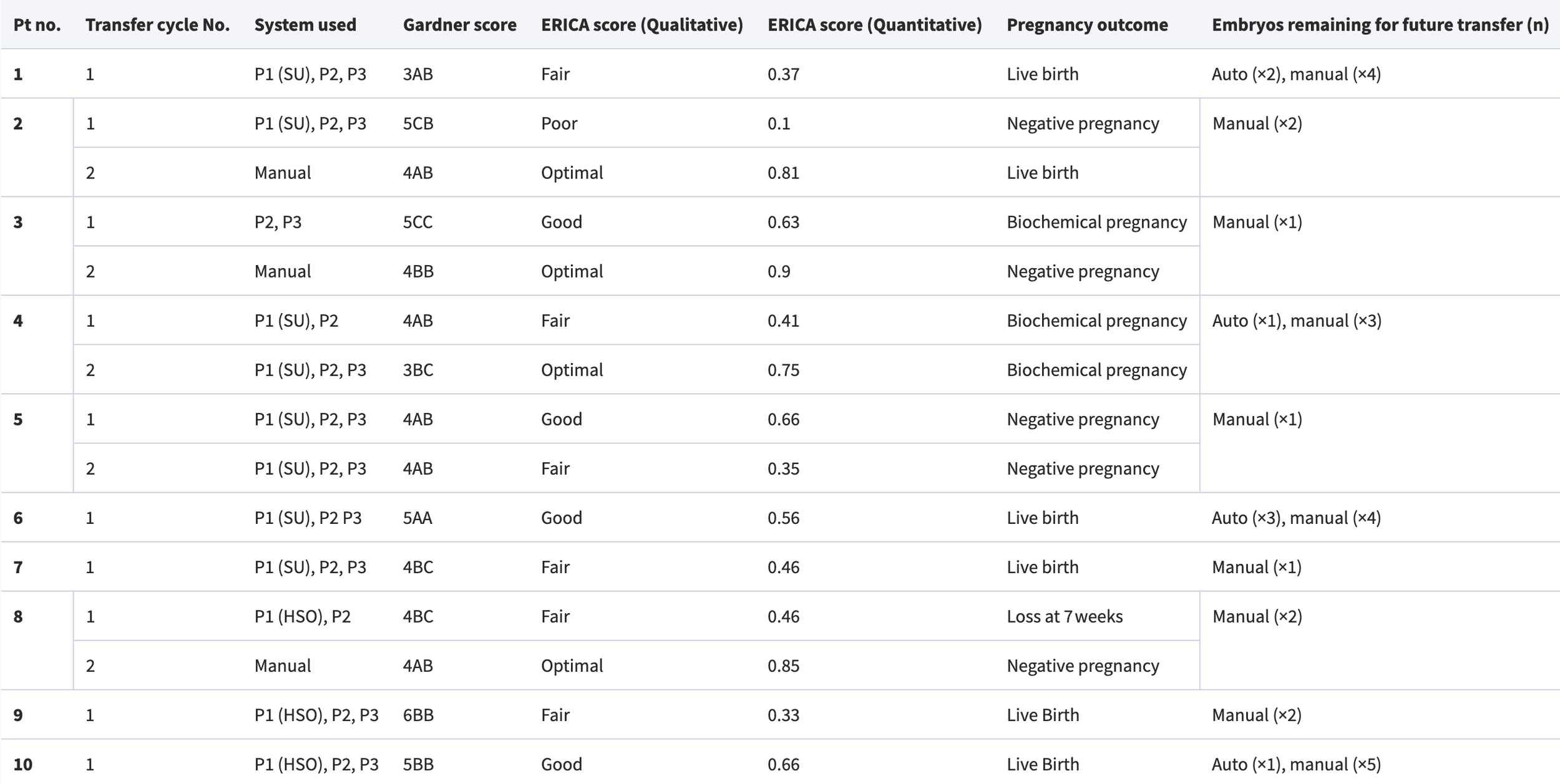
Transferred embryos (all vitrified and warmed) from both automated and manual treatments are depicted in Fig. 4, pre-vitrification, and post-warming. Detailed profiles including the assigned Gardner score (Gardner and Schoolcraft, 1999) and AI-assigned ERICA scores for the blastocysts (Chavez-Badiola et al., 2020) are provided in Table 5 along with clinical outcomes. Nine pregnancies were established in the automation group. Five pregnancies were delivered, and five healthy babies were born. Three pregnancies were biochemical, and one pregnancy ended in early loss at 7 weeks gestation (presence of a sac with no foetal heart activity). Three of the patients who did not achieve an ongoing pregnancy after transfer of an embryo from the automation treatment had a single vitrified–warmed blastocyst transfer from the manual treatment. One of the three patients has delivered a healthy baby. The other two patients did not become pregnant. All patients except one who had fertilization failure have remaining embryos from the manual treatment in cryostorage, while four of these patients have additional cryopreserved embryos from automated systems.
Figure 4.
Embryo morphology and transfer results. Embryo morphology was classified according to Gardner and Schoolcraft (1999) and by using the artificial intelligence assistant ERICA (Chavez-Badiola et al., 2020), which provides both a qualitative and quantitative score ranging between 0 and 1. Both Garnder and ERICA scores were assigned pre-vitrification. Negative pregnancy was defined as a blood beta hCG measurement lower than 20 IU/ml; biochemical pregnancy loss as decreasing serum or urinary beta hCG levels before ultrasound evaluation; early pregnancy loss as spontaneous loss before 10 weeks gestation. Scale bars equal ∼50 µm.
Video 1.
Operation of the Pearl 1 system for sperm preparation by Swim-Up (SU). A semen sample held in a standard cup is automatically aspirated and deposited at the bottom of a conical tube, then slowly overlaid with medium to initiate sperm preparation by direct swim-up. After 20 min, the system automatically collects the supernatant, completing the procedure.
Watch Video Here
Video 2.
Operation of the Pearl 1 system for sperm preparation by Horizontal Swim-Out (HSO). A six-axis robotic arm dispenses medium droplets in a dish and drags a pipette tip across them to create interconnecting channels in a set of four repeating patterns, creating a horizontal swim-out setup (see also Fig. 2). An operator then overlays the automatically configured droplets with mineral oil. The robotic arm then collects a small volume of semen from a standard cup and adds a predefined amount to each of four horizontal swim-out patterns in droplet #1 (per Fig. 2A). Onboard computer vision tracks the number of sperm cells per high-power field by observing sperm swimming in the last interconnecting channel. When the required concentration is achieved, the robotic arm severs the interconnecting channels, interrupting and completing the HSO process.
Video 3.
Pearl 2 system demonstration. This demonstration utilized cattle cumulus–oocyte complexes (COCs) in follicular fluid. Using an onboard camera and computer vision software, the system systematically scans the dish containing the follicular fluid and identifies putative COCs. Each COC is automatically aspirated through a fine pipette and deposited in washing medium into a separate dish.
Table 5. Single embryo transfer outcomes and the systems used to generate the embryos.
Pt, patient; P1, sperm preparation; P2, cumulus–oocyte complex (COC) retrieval and oocyte denudation; P3, ICSI; SU, swim-up; HSO, horizontal swim out. Negative pregnancy test was defined as beta hCG lower than 20 IU/ml. Biochemical pregnancy loss was defined as decreasing serum or urinary beta hCG levels, before an ultrasound evaluation. Early pregnancy loss was defined as spontaneous pregnancy loss before 10 weeks gestation. Definitions are based on (Kolte et al., 2015).
Discussion
We sought to establish the feasibility of automating the Day 0 gamete manipulation workflow in the IVF laboratory, specifically, oocyte retrieval, sperm preparation, and ICSI procedures. The results demonstrate that the automated protocols are feasible and, with human oversight and assistance, can produce acceptable fertilization and blastocyst formation rates. Moreover, transfer of embryos generated using these systems can lead to pregnancies and live births.
It should be noted that these prototype systems have been further developed and incorporated into a system called Automated Reproductive Assistant, or AURA, which is currently undergoing clinical testing in an IRB-approved study (ClinicalTrials.gov ID NCT06581068).
The automated robotic swim-up system (Pearl 1 SU) addresses the labour-intensive and variability-prone steps of manual sperm preparation protocols (Janssens and Cheung, 2009). The system performed precise layering of medium over semen and removed a predetermined volume of the upper layer of the swim-up after a specified time had elapsed. Processing by the robotic system produced highly motile final samples without seminal plasma carry-over or the need for centrifugation.
In the Pearl 1 HSO prototype, the dish was prepared partially automatically (medium was dispensed in the dish by the robotic arm and oil was dispensed by a human operator). This method required a small aliquot of the semen specimen and allowed sperm to swim out from semen-containing droplets through inter-connected channels into media droplets and eventually into a drop of PVP for use during ICSI. The HSO method required an average of 30.8 min per sample for complete sperm preparation, and the process is stopped by automated disruption of the channels when adequate numbers of sperm are observed in the final drop. This is visualized through an inverted microscope customized for automation.
Semen samples processed by microfluidic-based devices could be contaminated with seminal plasma carryover, and final samples require additional washing/centrifugation before use in conventional IVF to avoid detrimental effects on fertilization (Adolfsson et al., 2024). Semen samples prepared by the two Pearl 1 automated systems had no measurable trace of PSA by semi-quantitative testing (Millan et al., 2024), indicating the absence of seminal fluid. The presence of semen constituents in the final prepared sperm samples can hinder hyperactivation, acrosome reaction, and fertilization during IVF (Björndahl et al., 2005).
Avoiding centrifugation also avoids inducing DNA fragmentation in sperm cells (Muratori et al., 2019); however, this was not tested here. A significant limitation of these robotic systems is that they are not designed to handle severely oligozoospermic or asthenozoospermic samples or those with a combination of these abnormalities.
Follicular fluid was successfully searched by the AI-enabled, automated egg retrieval prototype (Pearl 2), and 92/110 or 84% of the oocytes were found successfully. Two oocytes of one patient were lost during these manipulations, and the process was relatively slow, requiring an average of 76 s per COC. Additionally, an embryologist received the follicular fluid tubes from the operating room, decanted the fluid in dishes, and placed the dish on the microscope stage before the robotic operation was started. The embryologist also checked the dishes initially to ensure that all COCs were noted and subsequently recovered by the robotic system. Following isolation, washing of the COCs was performed by the automated system, although this process required more time than would typically be expected for manual washing by an experienced embryologist (a mean of 1.8 min per COC), and dissection of the cumulus to remove blood clots or excessive granulosa cells was not possible. Denuding via enzymatic treatment and iterative pipetting was completed without damaging any oocytes; most oocytes had some remaining corona cells requiring intervention by embryologists for manual removal of the cells.
The automated ICSI system showed consistency as it determined a path for the injection needle, which was used in all injections (Mendizabal-Ruiz et al., 2025). The use of a piezo actuator for membrane breakage avoided distortion of the oocyte shape and potential cellular stress during injection (Fujii et al., 2020; Zander-Fox et al., 2024). The use of a single laser pulse to reduce the thickness of the zona pellucida at the site of injection aided the same effect.
Sperm selection during ICSI using AI software (SiD; Mendizabal-Ruiz et al., 2022) and immobilization using laser shots precisely directed at the centre of the sperm tail are both advantageous as they remove subjectivity from selection and variability in mechanical microneedle-mediated sperm immobilization (Mendizabal-Ruiz et al., 2025).
Speed was a limitation, as each injection required an average of 8.7 min, including sperm selection and immobilization (3.9 min), and oocyte handling and sperm placement (4.9 min); the manual sperm pick up and injection procedure was typically completed in ∼2 min. Extended exposure to HEPES-buffered media (Morgia et al., 2006) as well as potentially decreasing/fluctuating temperatures during manipulation can be detrimental during ICSI, thus the total time of manipulation must be carefully controlled and minimized. Speed of injection is expected to improve with further development of both the AI features and the robotics systems. It should also be acknowledged that an automated conventional insemination system remains to be developed and is important. Recent studies have shown that in non-male factor cases, ICSI does not improve cumulative live birth rates compared to conventional IVF (Wang et al., 2024; Berntsen et al., 2025) and may even be counterproductive (Paffoni et al., 2024). It will be interesting to see if some of the shortcomings of ICSI shown in these studies are related to variability in manual techniques and could be addressed by standardization through automation.
Fertilization and blastocyst formation outcomes for the automation group met competency levels based on the Vienna Consensus KPI (ESHRE Special Interest Group of Embryology and Alpha Scientists in Reproductive Medicine, 2017).
Pregnancy results were also within the expected range for the population of patients treated (ESHRE Special Interest Group of Embryology and Alpha Scientists in Reproductive Medicine, 2017; Vaiarelli et al., 2023). All pregnancies except one were achieved without preimplantation genetic testing for aneuploidy. Only one patient received a tested euploid embryo, and this led to a healthy live birth. A majority of those patients who did not become pregnant have additional embryos in cryostorage available for transfer either from the manual group or from the automated group. Thus, cumulative pregnancy rates are expected to remain in the acceptable range. There were three biochemical pregnancies and one loss at 7 weeks (foetal sac with no heartbeat) in this subset of nine pregnancies.
Taken together, the laboratory and clinical outcomes suggest that automated handling of oocytes, sperm, and embryos with oversight by an experienced embryologist is feasible and does not have detrimental effects on development potential beyond those that may result from manual processes.
These findings confirm that the use of multiple automated systems to perform procedures on Day 0 of development is feasible and compatible with embryo viability, establishing healthy pregnancies, and delivery of healthy babies. The results highlight the potential to standardize procedures and reduce operator-dependent variability and errors. Ultimately, standardization is expected to help improve laboratory efficiency and outcomes (Alikani and Campbell, 2025). As improvements in hardware, image processing, and AI algorithms are made, a fully automated workflow can be envisioned that is capable of high-volume case management. Scalability is a significant challenge and remains to be demonstrated.
This was a limited pilot series using prototype automated systems. Although automation is poised to reduce manual workload, currently, it requires advanced operator training in multiple disciplines for troubleshooting. Indeed, it should be noted that the Pearl systems evaluated in this study did not achieve complete autonomy, defined as the ability to perform procedures without any operator input, intervention, or supervision. Moreover, automated devices/equipment and AI-driven technologies present a unique set of ethical and other challenges (Mennella et al., 2024) as well as higher initial investment costs, which can be barriers to adoption.
In the light of the continuum proposed by Provoost et al. (2014) for distinguishing between experimental, innovative, and established treatments, these Day 0 automation procedures appear to fit most appropriately in the ‘innovative’ phase. This assessment aligns with the ASRM Practice Committee’s criteria for innovation, which emphasizes that novel interventions should demonstrate adequate evidence of effectiveness, appropriate informed consent processes, and ongoing outcome monitoring (Ethics Committee of the American Society for Reproductive Medicine, 2021). Proof of principle in human subjects is established here, and the safety data are reassuring. However, the aggregate body of evidence is still limited. Our findings thus support continued, methodical integration of automation, with improvements, in a research-monitoring framework.
As these technologies evolve and are further optimized, they should be viewed not just as new technology but as a strategic approach to maintaining reproducibility and reliability and a way to streamline workflows to meet the growing demand for ART.
Acknowledgements
-
Individual participant data that underlie the results reported in this article (including text, tables, figures, and appendices), after de-identification, will be made available to researchers who provide a methodologically sound proposal for use in achieving the aims outlined in the approved proposal. Data will be available beginning 3 months and ending 36 months following article publication. Proposals may be submitted up to 36 months post-publication and should be directed to jacques@conceivable.life. Access will be granted upon completion of a data access agreement.
-
A.C.-B.: Conceptualization, Formal Analysis, Investigation, Project Administration, Supervision, Writing—Original Draft, Writing—Review & Editing. G.M.-R.: Conceptualization, Investigation, Methods, Project Administration, Software, Visualization, Writing—Original Draft, Writing—Review & Editing. A.F.-S.F.: Conceptualization, Data Curation, Formal Analysis, Investigation, Methods, Project Administration, Validation, Visualization, Writing—Original Draft, Writing—Review & Editing. N.C.-B.: Conceptualization, Investigation, Methods, Project Administration, Validation, Writing—Original Draft, Writing—Review & Editing. A.M.: Conceptualization, Project Administration, Supervision, Writing—Review & Editing. M.A.: Conceptualization, Data Curation, Formal Analysis, Project Administration, Supervision, Visualization, Writing—Original Draft, Writing—Review & Editing. G.S.: Data Curation, Formal Analysis, Visualization, Writing—Original Draft, Writing—Review & Editing. C.M.: Investigation, Methods, Validation, Writing—Review & Editing. E.H.-M.: Investigation, Methods, Software, Validation, Writing—Review & Editing. R.V.-M.: Methods, Software. Victor Medina: Investigation, Software, Writing—Review & Editing. E.M.: Investigation, Methods, Validation, Writing—Review & Editing. A.V.A.: Investigation, Validation, Visualization, Writing—Review & Editing. V.O.-H.: Software, Writing—Review & Editing. F.A.: Investigation, Software, Validation, Writing—Review & Editing. A.A.L.: Methods, Software, Validation. M.A.: Investigation, Methods, Validation, Writing—Review & Editing. Q.M.-A.: Investigation, Methods, Validation, Writing—Review & Editing. J.G.E.F.: Investigation, Software, Validation, Writing—Review & Editing. L.M.C.O.: Investigation, Writing—Review & Editing. C.P.B.: Data Curation, Investigation, Visualization, Writing—Review & Editing. D.J.S.-G.: Investigation, Writing—Review & Editing. J.C.: Conceptualization, Data Curation, Formal Analysis, Methods, Project Administration, Supervision, Writing—Original Draft, Writing—Review & Editing.
-
No specific funding was sought for the study. The study was sponsored by Conceivable Life Sciences, New York, NY, USA.
-
A.C.-B. is an employee, shareholder, and company officer at Conceivable Life Sciences, and also holds shares in IVF 2.0. G.M.-R. is a stock option holder at Conceivable Life Sciences and has received consulting fees from both Conceivable Life Sciences and IVF 2.0. A.F.-S.F. is an employee and stock option holder at Conceivable Life Sciences. N.C.-B. is a stock option holder at Conceivable Life Sciences and has received consulting fees from the company and he is also an employee, shareholder, and company officer at Embryotools. A.M. is a shareholder and company officer at Conceivable Life Sciences, and holds shares in TMRW Life Sciences. M.A. is a stock option holder at Conceivable Life Sciences, and has received consulting fees and travel support from the company; she has also received consulting fees from TMRW Life Sciences. G.S. is an employee and stock option holder at Conceivable Life Sciences and has received consulting fees from IVF 2.0. C.M. is an employee and stock option holder at Conceivable Life Sciences. E.H.-M. is an employee and stock option holder at Conceivable Life Sciences. R.V.-M. is an employee and stock option holder at Conceivable Life Sciences and has received consulting fees from IVF 2.0. V.M. is an employee and stock option holder at Conceivable Life Sciences. E.M. is an employee of Embryotools. A.V.A. has no conflicts of interest to declare. V.O.-H. is an employee and stock option holder at Conceivable Life Sciences and has received consulting fees from IVF 2.0. F.A.-G. is an employee and stock option holder at Conceivable Life Sciences. A.Á.L. is an employee and stock option holder at Conceivable Life Sciences. M.A. is an employee of Embryotools. Q.M.-A. is an employee of Embryotools. J.G.E.F. is an employee and stock option holder at Conceivable Life Sciences. L.M.C.O. has no conflicts of interest to declare. C.P.B. is an employee and stock option holder at Conceivable Life Sciences. D.J.S.-G. has no conflicts of interest to declare. J.C. is an employee, shareholder, stock option holder, and company officer at Conceivable Life Sciences and holds shares in IVF 2.0, Althea Science, TMRW Life Sciences, Kindbody, and Reproductive Healthcare. Furthermore, A.C.-B., G.M.-R., A.F.-S.F., A.M., A.Á.L., and J.C. declare inventorship on US patent 12,349,940 B2 protecting automated oocyte denudation. A.C.-B., G.M.-R., A.F.-S.F., C.M., R.V.-M., V.O.-H., E.H.-M., V.M., A.M., and J.C. declare inventorship on US patent 12,245,793 B2 protecting robotic handling systems for IVF laboratories. A.C.-B., G.M.-R., A.F.-S.F., C.M., G.S., V.M., J.C., and A.M. declare inventorship on US patent 12,178,475 B1 protecting automated sperm preparation. A.C.-B., G.M.-R., A.M., and J.C. declare inventorship on US patent 12,180,441 B1 protecting laboratory workflow automation. A. C.-B., G.M.-R., A.F.-S.F., A.M., C.M., R.V.-M., V.O.-H., N.C.-B., A.Á.L., and J.C. declare inventorship on US patent 12,268,418 B2 protecting automated oocyte preparation. A.C.-B., G.M.-R., R.V.-M., V.O.-H., N.C.-B., E.H.-M., A.M., J.C., and A.F.-S.F. declare inventorship on US patent 12,310,625 B2 protecting automated ICSI. A.C.-B., G.M.-R., A.F.-S.F., C.M., V.O.-H., and A.M. declare inventorship on US patent 12,253,516 B2 protecting optical systems for IVF automation. A.C.-B., G.M.-R., A.M., J.C., A.F.-S.F., C.M., R.V.-M., V.O.-H., G.S., N.C.-B., and J.G.E.F. declare inventorship on US patent 12,226,125 B2 protecting automated vitrification. A.C.-B., G.M.-R., C.M., R.V.-M., V.O-H., N.C.-B.,V.M., and J.C. declare inventorship on US patent US 12,478,405 B2 protecting automated dish-based sperm preparation. A.C.-B., G.M.-R., C.M., N.C.-B., M.A., A.M., and J.C. declare inventorship on a pending patent application protecting automated dish preparation technology. A.C.-B., G.M.-R., N.C.-B., and J.C. declare inventorship on a pending patent application protecting automated oocyte denudation. N.C.-B., J.C., A.M., E.H.-M., and A.C.-B. declare inventorship on a pending patent application protecting automated vitrification systems. A.C.-B., G.M.-R., V.O.-H., and J.C. declare inventorship on a pending patent application protecting cumulus-oocyte complex detection technology.
-
Adolfsson E, Ingberg J, Ingberg E, Bohlin T. Clinical validation and experiences of the microfluidics sperm preparation device ZyMot™. Reproductive Biomedicine Online. 2024;48:103995.
Agarwal A, Henkel R, Huang C, Lee M. Automation of human semen analysis using a novel artificial intelligence optical microscopic technology. Andrologia. 2019;51:e13440.
Alikani M, Campbell A. Shaping the future of the IVF laboratory: standardization for more predictable outcomes. Reproductive Biomedicine Online. 2025;50:104854.
Alikani M, Go KJ, McCaffrey C, McCulloh DH. Comprehensive evaluation of contemporary assisted reproduction technology laboratory operations to determine staffing levels that promote patient safety and quality care. Fertility and Sterility. 2014;102:1350–1356.
Baldini D, Baldini A, Silvestris E, Vizziello G, Ferri D, Vizziello D. A fast and safe technique for sperm preparation in ICSI treatments within a randomized controlled trial. Reproductive Biology and Endocrinology. 2020;18:88.
Berntsen S, Zedeler A, Nøhr B, Rønn Petersen M, Grøndahl ML, Andersen LF, Løssl K, Løkkegaard E, Englund AL, Vestergaard Gabrielsen A, et al. IVF versus ICSI in patients without severe male factor infertility: a randomized clinical trial. Nature Medicine. 2025;31:1939–1948.
Björndahl L, Mohammadieh M, Pourian M, Söderlund I, Kvist U. Contamination by seminal plasma factors during sperm selection. Journal of Andrology. 2005;26:170–173.
Chavez-Badiola A, Flores-Saiffe-Farías A, Mendizabal-Ruiz G, Drakeley AJ, Cohen J. Embryo Ranking Intelligent Classification Algorithm (ERICA): artificial intelligence clinical assistant predicting embryo ploidy and implantation. Reproductive Biomedicine Online. 2020;41:585–593.
Costa-Borges N, Farías AFS, Mendizabal G, Hernandez EG, Acacio M, Matia-Algué Q, Mestres E, Barragan CP, Carreon DSM, Calderon G. Laser immobilization of spermatozoa for intracytoplasmic sperm injection: an exploratory and safety study in the mouse model. Fertility and Sterility. 2024;122:e342.
Costa-Borges N, Matia-Algué Q, Coello A, Mestres E, Acacio M, Flores-Saiffe Farías A, Castello C, Gallardo M, Chavez-Badiola A, Marco-Jiménez F, et al. Preclinical validation of fast oocyte vitrification and warming protocols with comparable efficiencies to a standard method. Human Reproduction. 2025;40:1066–1076.
Costa-Borges N, Munné S, Albó E, Mas S, Castelló C, Giralt G, Lu Z, Chau C, Acacio M, Mestres E, et al. First babies conceived with automated intracytoplasmic sperm injection. Reproductive Biomedicine Online. 2023;47:103237.
De Martin H, Cocuzza MS, Tiseo BC, Wood GJ, Miranda EP, Monteleone PA, Soares JM, Serafini PC, Srougi M, Baracat EC. Positive rheotaxis extended drop: a one-step procedure to select and recover sperm with mature chromatin for intracytoplasmic sperm injection. Journal of Assisted Reproduction and Genetics. 2017;34:1699–1708.
ESHRE Special Interest Group of Embryology and Alpha Scientists in Reproductive Medicine. The Vienna consensus: report of an expert meeting on the development of ART laboratory performance indicators. Reproductive Biomedicine Online. 2017;35:494–510.
Ethics Committee of the American Society for Reproductive Medicine. Moving innovation to practice: an Ethics Committee opinion. Fertility and Sterility. 2021;116:331–336.
Ferrer Buitrago M, Ferrer-Robles E, Muñoz-Soriano P, Antequera-Durán V, Gómez-Giménez B, Bataller-Sánchez J, Barberà-Alberola A, Calatayud-Lliso C, Ruiz-Jorro M. ICSI outcomes after using in-situ microfluidics of fluidic walls versus density gradient centrifugation: a prospective non-inferiority comparative pilot study in sibling oocytes. Human Reproduction. 2023;38:dead093-585.
Forbrig M, Peirce K, Copeland C, Natalwala J, Chapple V, Liu Y. Risk factors for mismatches in the ART laboratory: an analysis of 73,719 electronic witnessing points. Reproductive Biomedicine Online. 2025;50:104500.
Fujii Y, Endo Y, Mitsuhata S, Hayashi M, Motoyama H. Evaluation of the effect of piezo-intracytoplasmic sperm injection on laboratory, clinical, and neonatal outcomes. Reproductive Medicine and Biology. 2020;19:198–205.
Gardner DK, Schoolcraft WB. Culture and transfer of human blastocysts. Current Opinion in Obstetrics and Gynecology. 1999;11:307–311.
Janssens PM, Cheung KS. Approaching risk analysis and risk management in the fertility laboratory and semen bank. International Journal of Andrology. 2009;32:656–665.
Jiang VS, Bormann CL. Artificial intelligence in the in vitro fertilization laboratory: a review of advancements over the last decade. Fertility and Sterility. 2023;120:17–23.
Kolte A, Bernardi L, Christiansen O, Quenby S, Farquharson R, Goddijn M, Stephenson M. Terminology for pregnancy loss prior to viability: a consensus statement from the ESHRE early pregnancy special interest group. Human Reproduction. 2015;30:495–498.
Lattin MT, Djandji AS, Kronfeld MT, Samsel T, Ling R, Ciskanik M, Sadowy S, Forman EJ, Williams Z. Development and validation of an automated robotic system for preparation of embryo culture dishes. Fertility and Sterility. 2024;122:297–303.
Lu Z, Zhang X, Leung C, Esfandiari N, Casper RF, Sun Y. Robotic ICSI (intracytoplasmic sperm injection). IEEE Transactions on Biomedical Engineering. 2011;58:2102–2108.
Mendizabal-Ruiz G, Chavez-Badiola A, Figueroa IA, Nuño VM, Farías AFS, Valencia-Murillo R, Drakeley A, Garcia-Sandoval JP, Cohen J. Computer software-assisted real-time single sperm selection associated with fertilization and blastocyst formation. Reproductive Biomedicine Online. 2022;45:703–711.
Mendizabal-Ruiz G, Chavez-Badiola A, Hernández-Morales E, Valencia-Murillo R, Ocegueda-Hernández V, Costa-Borges N, Mestres E, Acacio M, Matia-Algué Q, Farías AFS, et al. A digitally controlled, remotely operated ICSI system: case report of the first live birth. Reproductive Biomedicine Online. 2025;50:104943.
Mennella C, Maniscalco U, De Pietro G, Esposito M. Ethical and regulatory challenges of AI technologies in healthcare: a narrative review. Heliyon. 2024;10:e26297.
Millan C, Pizano Parra AA, Mendizabal G, Medina V, Saiffe Farías AFS, Barragan CP, Martinez Carreon DS, Valadez A, Aleriano K, Silvestri G, et al. Development and preliminary testing of a fully automated semen preparation robot. Fertility and Sterility. 2024;122:e122.
Morgia F, Torti M, Montigiani M, Piscitelli C, Giallonardo A, Schimberni M, Giannini P, Sbracia M. Use of a HEPES-buffered medium in intracytoplasmic sperm injection procedures is detrimental to IVF outcomes. Fertility and Sterility. 2006;85:1415–1419.
Muratori M, Tarozzi N, Carpentiero F, Danti S, Perrone FM, Cambi M, Casini A, Azzari C, Boni L, Maggi M, et al. Sperm selection with density gradient centrifugation and swim-up: effect on DNA fragmentation in viable spermatozoa. Scientific Reports. 2019;9:7492.
Paffoni A, Vitagliano A, Corti L, Somigliana E, Viganò P. Intracytoplasmic sperm injection versus conventional in vitro insemination in couples with non-male infertility factor: real-world analysis of the HFEA registry. Journal of Translational Medicine. 2024;22:687.
Palmer GA, Chavez-Badiola A, Valencia-Murillo R, Harvey SC, Mendizabal-Ruiz G, Farías AFS, Paredes O, Griffin DK. Can AI-guided feedback improve embryologists’ selection of euploid embryos based on morphology alone? Reproductive Biomedicine Online. 2025;51:104990.
Practice Committee of the American Society for Reproductive Medicine. Prevention of moderate and severe ovarian hyperstimulation syndrome: a guideline. Fertility and Sterility. 2024;121:230–245.
Provoost V, Tilleman K, D’Angelo A, De Sutter P, de Wert G, Nelen W, Pennings G, Shenfield F, Dondorp W. Beyond the dichotomy: distinguishing experimental, innovative, and established treatment. Human Reproduction. 2014;29:413–417.
Roy TK, Brandi S, Peura TT. Gavi-automated vitrification instrument. In: Nagy Z, Varghese A, Agarwal A (eds). Cryopreservation of Mammalian Gametes and Embryos: Methods and Protocols. New York: Springer; 2017:261–277.
Ruiz-Jorro M, Bataller-Sánchez J, Barberá-Alberola A, Gómez-Giménez B, Moliner-Aguilar V, Calatayud-Lliso C, Ferrer-Buitrago M. In situ microfluidics of fluidic walls: a novel deviceless approach for sperm selection in the same ICSI dish. Human Reproduction. 2023;38:dead093-426.
Sakkas D, Barrett CB, Alper MM. Types and frequency of non-conformances in an IVF laboratory. Human Reproduction. 2018;33:2196–2204.
Tiegs AW, Scott RT. Evaluation of fertilization, blastocyst development, and implantation rates according to ICSI operator experience. Reproductive Biomedicine Online. 2020;41:19–27.
Vaiarelli A, Zacà C, Spadoni V, Cimadomo D, Conforti A, Alviggi C, Palermo R, Bulletti C, De Santis L, Pisaturo V, et al. Clinical and laboratory key performance indicators in IVF: a consensus statement. Journal of Assisted Reproduction and Genetics. 2023;40:1479–1494.
Veiga E, Olmedo C, Sánchez L, Fernández M, Mauri A, Ferrer E, Ortiz N. Recalculating staffing requirements for a modern ART laboratory. Human Reproduction. 2022;37:1774–1785.
Wang Y, Li R, Yang R, Zheng D, Zeng L, Lian Y, Zhu Y, Zhao J, Liang X, Li W, et al. Intracytoplasmic sperm injection versus conventional IVF for couples with non-severe male factor infertility. The Lancet. 2024;403:924–934.
Yang G-Z, Cambias J, Cleary K, Daimler E, Drake J, Dupont PE, Hata N, Kazanzides P, Martel S, Patel RV, et al. Medical robotics: regulatory, ethical, and legal considerations for increasing autonomy. Science Robotics. 2017;2:eaam8638.
Zander-Fox D, Green M, Watson K, Turner R, Bakos HW, Foo J, Pacella-Ince L, Caddy M, McPherson NO, Rombauts L. Improved fertilization and embryo quality rates with PIEZO–ICSI compared with conventional ICSI: a sibling oocyte split multicenter trial. Fertility and Sterility. 2024;121:971–981.