Investigating Phototoxicity of Optical Coherence Tomography (OCT) Imaging in Porcine and Human Sperm
Aquilina et al., Reproductive BioMedicine Online (RMBO), 2025
Table Of ContentsIntroduction
Materials and Methods
Results
Discussion
Acknowledgements
Abstract
Research questionDoes exposure to near-infra-red optical coherence tomography (OCT) laser radiation induce phototoxic effects in porcine and human spermatozoa?
DesignComputer-assisted sperm analysis (CASA) was used to determine whether OCT laser illumination at or above levels typically used for imaging alters sperm motility. Flow cytometry was used to determine the impact of irradiation on sperm acrosome reaction status, DNA fragmentation, and membrane integrity. Additionally, in-vitro time-lapse OCT imaging of a porcine cumulus–oocyte complex with irradiated spermatozoa was performed to determine whether irradiated spermatozoa interact with and penetrate the cumulus oophorus. Finally, human spermatozoa were irradiated and analysed using CASA and flow cytometric techniques.
ResultsAll irradiated samples showed no significant difference in sperm DNA damage or CASA sperm motility parameters, including average path velocity, straight line velocity or curvilinear velocity, compared with their matched manipulation control, suggesting that sample irradiation did not compromise sperm viability, even when using an optical power of more than one order of magnitude greater than that typically required to image embryos. Proof-of-concept OCT imaging suggested that motility of irradiated spermatozoa during cumulus interaction was not affected by radiation.
ConclusionsNo significant effect on the kinetics of boar and human spermatozoa was observed following near-infra-red OCT laser radiation. Future work will investigate the fertilization process and embryo development following near-infra-red OCT laser radiation.
KEY WORDSInfra-red • Optical coherence tomography • Phototoxicity • Sperm DNA damage
Introduction
In reproductive medicine, time-lapse imaging systems (TLS) have provided new insights into the morphological processes of fertilization and embryo development (Castellò et al., 2016; Kovacs, 2014). Their clinical safety is widely accepted (Kirkegaard et al., 2012; Nakahara et al., 2010), and algorithms have been developed to identify the ideal morphokinetic parameters for achieving successful pregnancy outcomes (Chamayou et al., 2013; Cruz et al., 2012; Dal Canto et al., 2012; Petersen et al., 2016; Storr et al., 2018). However, current TLS do not offer intrinsic depth discrimination capability, making it difficult to resolve structures located at different depths. Optical coherence tomography (OCT) is an imaging technique that utilizes low-coherence interferometry for non-invasive generation of three-dimensional (3D) cross-sectional images of live cells and tissues of interest, both in vitro and in vivo (Alam and Poddar, 2022; Cernat et al., 2012; Holmes, 2009; Huang et al., 1991; Popescu et al., 2011; Sepehr et al., 2008). The 3D nature of OCT imaging therefore offers a novel perspective for time-lapse monitoring of early embryonic progression, providing a non-invasive method of visualizing morphokinetic events during development.
OCT is not limited to ‘snapshot’ imaging of cell positioning. OCT images can also be processed to understand cellular and subcellular movements by processing time-resolved images acquired at the same spatial position in the sample and interrogating the variability of the signal over time – high variability corresponds to increasing movement of cellular and subcellular structures (Apelian et al., 2016; Ren et al., 2024). As active motions are not present in dead cells (i.e. thermal/Brownian motion background alone), OCT imaging at regular time intervals allows for the measurement of cellular motility and vitality while providing additional contrast on tissue morphology (Apelian et al., 2016; Kohlfaerber et al., 2022). In a reproductive context, while sperm cells are too small to be resolved directly by OCT, during fertilization, they interact with larger cells which are detectable by OCT. Thus, it may be possible to use OCT to study not only post-fertilization embryonic development, but also the fertilization process itself. In particular, the ability to observe changes occurring within the cumulus complex potentially offers new monitoring tools to help understand and diagnose fertility problems in couples presenting for assisted reproduction.
However, while progress has been made in the use of OCT to visualize embryonic structures at the blastocyst stage (Masuda et al., 2023) and throughout embryonic development (Karnowski et al., 2017), no studies to date have addressed the safety of OCT for gamete health, or the ability of OCT to detect perifertilization events such as sperm interactions with cumulus cells. Safe exposure of oocytes and pre-implantation embryos to light exposure has been discussed in a few papers (Khodavirdilou et al., 2021; Ottosen et al., 2007), although these focused on visible light exposure in the context of conventional microscopy. Safe exposure of oocytes and embryos to scanned near-infra-red light in OCT has been claimed in two other papers (Fluks et al., 2022; Karnowski et al., 2017), but these claims are only supported by observation of the first cellular division at cleavage stage. In addition, a study by Takae et al. (2017) analysed the safety of OCT examination on mouse ovarian tissue, and demonstrated that OCT had no effect on reproductive outcomes, including fertilization rate and blastocyst rate, nor was it associated with increased rates of birth defects.
No such report was found on the effect of OCT light exposure on spermatozoa. This understanding is important as spermatozoa are particularly prone to DNA damage due to their limited DNA repair ability after spermiogenesis (García-Rodríguez et al., 2018). Although low levels of unrepaired sperm DNA damage can be repaired in the zygote following fertilization (Ménézo et al., 2010), if sperm DNA damage exceeds the oocyte's reparative ability, there is increased risk of implantation failure, miscarriage, abnormal fetal development, and birth defects in offspring (González-Marín et al., 2012; Vasilyeva et al., 2020; Wyck et al., 2018). Moreover, spermatozoa, when in vivo, are thermotactic (Bahat and Eisenbach, 2010; Rodríguez-Gil, 2019), and thus not only their genomic integrity but also their behaviour and motility patterns could potentially be affected by infra-red illumination.
As such, there is a critical need to establish certain key factors before OCT can be used as a tool to study fertilization: firstly, whether OCT illumination alters sperm motility, viability or DNA fragmentation; secondly, whether spermatozoa subjected to OCT illumination remain competent to interact with and penetrate the cumulus oophorus; and thirdly, whether the cumulus interaction is itself visualizable by OCT methods. Therefore, this study analysed boar and human sperm parameters following exposure to various irradiation conditions, with the aim of providing an understanding of the genotoxic impact of broadband infra-red light on spermatozoa, and also visualizing the motility effects of sperm interactions with the cumulus during fertilization. Porcine spermatozoa were selected as a translational model of human spermatozoa, while providing an ethically favourable and cost-effective approach for initial screening. Finally, given that boar spermatozoa are more resistant to damage than human spermatozoa (Enciso et al., 2011), the effects of intra-red irradiation were examined in human donor samples to confirm the potential for clinical relevance, and ensure that the findings translate directly to the target species for assisted reproductive applications.
Materials and Methods
In initial experiments, the authors focused on determining how different OCT scanning protocols affect sperm function in relation to vitality, motility and acrosome status. Specifically, protocols were designed to study: (i) the impact of varying the total amount of energy received by the sample, while keeping the irradiation frequency constant (Protocol A); (ii) the impact of varying the irradiation frequency (i.e. the time between two successive exposures of spermatozoa) at a constant dose level (Protocol B); and (iii) whether transient exposure to higher frequency irradiation for part of the scan had a deleterious effect (Protocol C).
Semen analysisBoar semen samples were received in semen extender (for commercial artificial insemination) from JSR Genetics (UK). Semen extenders are media additives that prolong the life of sperm cells to aid logistics of shipping and distribution in commercial livestock production. While the exact composition of the extender is proprietary and is not disclosed by the company, this will not have affected the within-sample comparisons of the effect of infra-red irradiation in this study. The iSperm automated semen analysis device (Aidmics Biotechnology Co., China) was used for sperm kinetic evaluation following irradiation. Boar sperm concentration and motility (progressive, non-progressive and non-motile) were assessed according to the World Health Organization (WHO) 2010 criteria via the manual method (WHO, 2010). Human semen samples were collected by masturbation on the day of testing, and placed in an incubator at 37°C to allow for liquefaction. Human sperm concentration, motility and vitality were evaluated according to the WHO 2010 criteria via the manual method (WHO, 2010). Sperm motility was assessed by placing 2 × 10-µl drops of semen on opposite sides of a microscope slide, and covering using 22 × 22-mm coverslips. Sperm motility was assessed using a x40 phase objective (x400 total magnification), and the percentages of progressive motility, non-progressive motility and immotility were recorded. Two hundred sperm cells were counted in each drop of semen, and the average of the replicate results was compared with the acceptable differences criteria. If the criteria were not met, another 200 sperm cells were counted using a freshly prepared slide, and a third count was performed. Vitality was assessed using 0.5% Eosin Y in 0.9% sodium chloride (Gurr, 34197). Five microlitres of semen was mixed with 5 µl of the dye on a slide. A 22 × 22-mm coverslip was placed appropriately, and the semen was examined under a brightfield microscope. Colourless cells were counted as viable while pink stained cells were counted as non-viable. Two hundred sperm cells were counted. Another slide was assessed for vitality, with another 200 sperm cells counted; the average was taken for comparison with the acceptable differences criteria indicated by WHO (2010). If the criteria were not met, another 200 sperm cells were counted using a freshly prepared slide, and a third count was performed.
The LensHooke X1 PRO automated semen analysis system (Bonraybio, China) was used for further evaluation of sperm kinetics following irradiation. This system covers widely used measures of sperm motility, including average path velocity (VAP), straight line velocity (VSL) and curvilinear velocity (VCL) (Mortimer, 2000).
Flow cytometry analysisThe sperm chromatin structure assay (SCSA) was used for measuring the sperm DNA fragmentation index (DFI). Flow cytometry analysis was performed using a BD Accuri C6 Plus flow cytometer equipped with a 488-nm, 50-mW solid-state laser (BD Biosciences, UK). BD Accuri version 1.34.1 was used for data analysis and plot generation. The SCSA protocol was followed as described elsewhere (Evenson, 2013). Briefly, semen samples were diluted to 1–2 × 106 sperm/ml using TNE buffer. The samples were treated with an acid detergent solution for 30 s, followed by staining with an acridine orange solution to allow for the measurement of intact and fragmented DNA. Acridine orange exhibits differential binding to double-stranded and single-stranded DNA, resulting in a metachromatic shift from green to red fluorescence. A total of 15,000 sperm events per sample (5000 sperm cells per triplicate) were analysed using the flow cytometer.
Sperm viability and acrosome reactivity were evaluated using a staining solution composed of 1 μg/ml peanut agglutinin-fluorescein isothiocyanate and 1.5 μM propidium iodide in phosphate-buffered saline (Robles and Martínez-Pastor, 2013). The sperm sample was diluted in the staining solution to achieve a concentration of 1–2 × 106 sperm cells/ml, and analysed using a BD Accuri C6 Plus flow cytometer.
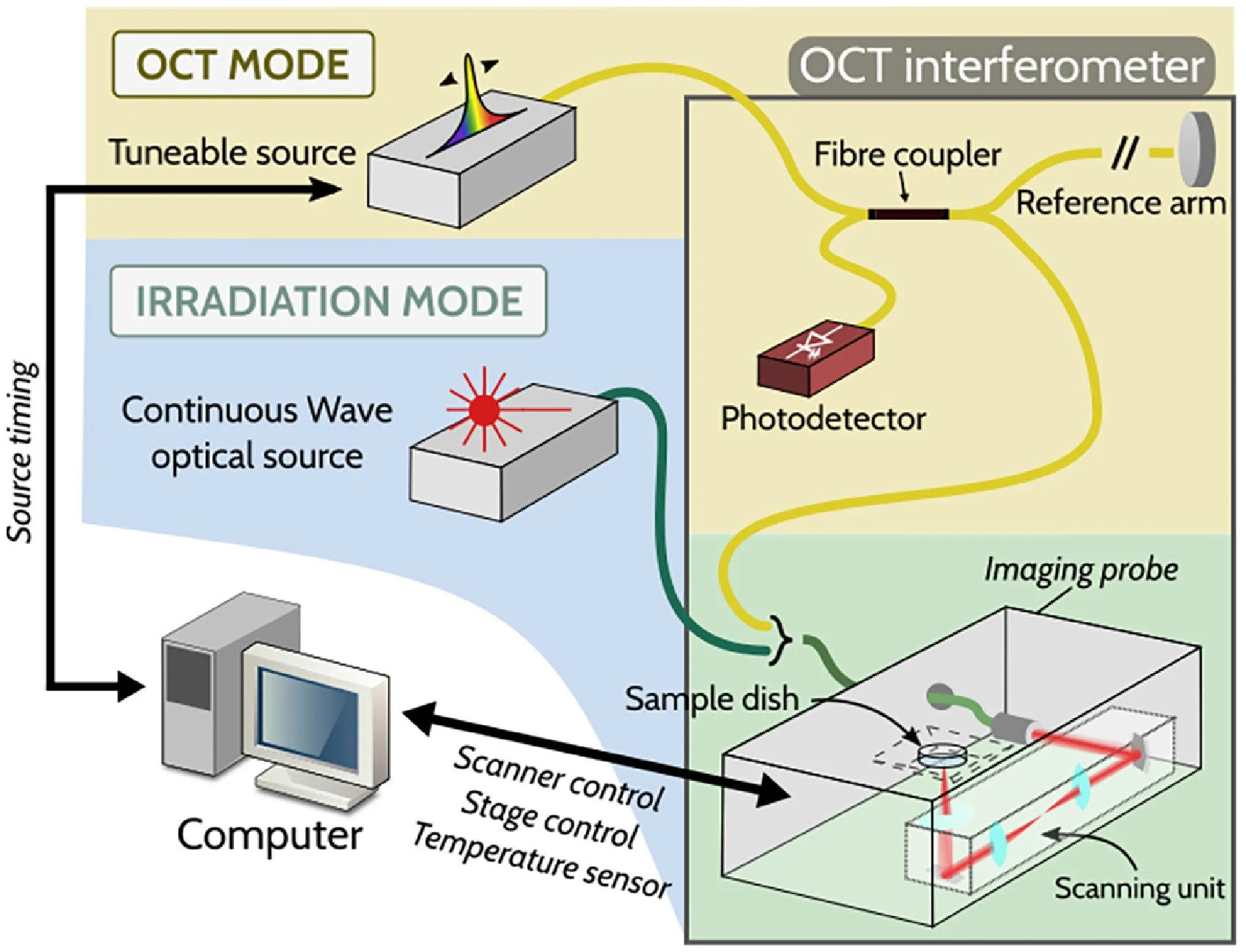
Optical system for semen irradiation and OCT imagingThe optical system used for semen irradiation and OCT imaging is presented in Figure 1. The system can be used in two modes as indicated by labels and coloured backgrounds. First, the ‘irradiation mode’ (shown on a blue background) aimed to mimic the operation of a point-scanning OCT system, without performing imaging. Light from a continuous wave optical source (MOPA-SLD; Superlum, Ireland) emitting at a wavelength of 1078 nm with a 20-nm bandwidth was coupled into an optical fibre, which was inserted into the imaging probe. Inside the imaging probe, the beam was coupled in air and scanned on to the sample using two scanning mirrors (SMX and SMY) (6215H; Novanta, USA). The scanned light travelled through an imaging lens and a wedged glass window, reaching the sample dish placed outside the imaging probe on top of the window. Second, the ‘OCT mode’ (shown on a yellow background) allowed for OCT imaging, employing a 200-kHz tuneable source (AXP50125-3; Axsun–Excelitas Technologies, USA) coupled to a fibre coupler which split light between the imaging probe and a reference arm (fibre array, not shown). Interference spectra were detected by a balanced photodetector (PDB471C; Thorlabs, USA).
Figure 1 Schematic diagram of the optical set-up for optical coherence tomography (OCT) imaging and irradiation experiments. The set-up comprises two modes: irradiation mode (represented on blue background) and OCT mode (represented on yellow background). The OCT interferometer consists of a fibre coupler that splits light from the tuneable laser source into the sample arm and reference arm. The light in the sample arm interacts with the specimen, and is collected back through the same path.
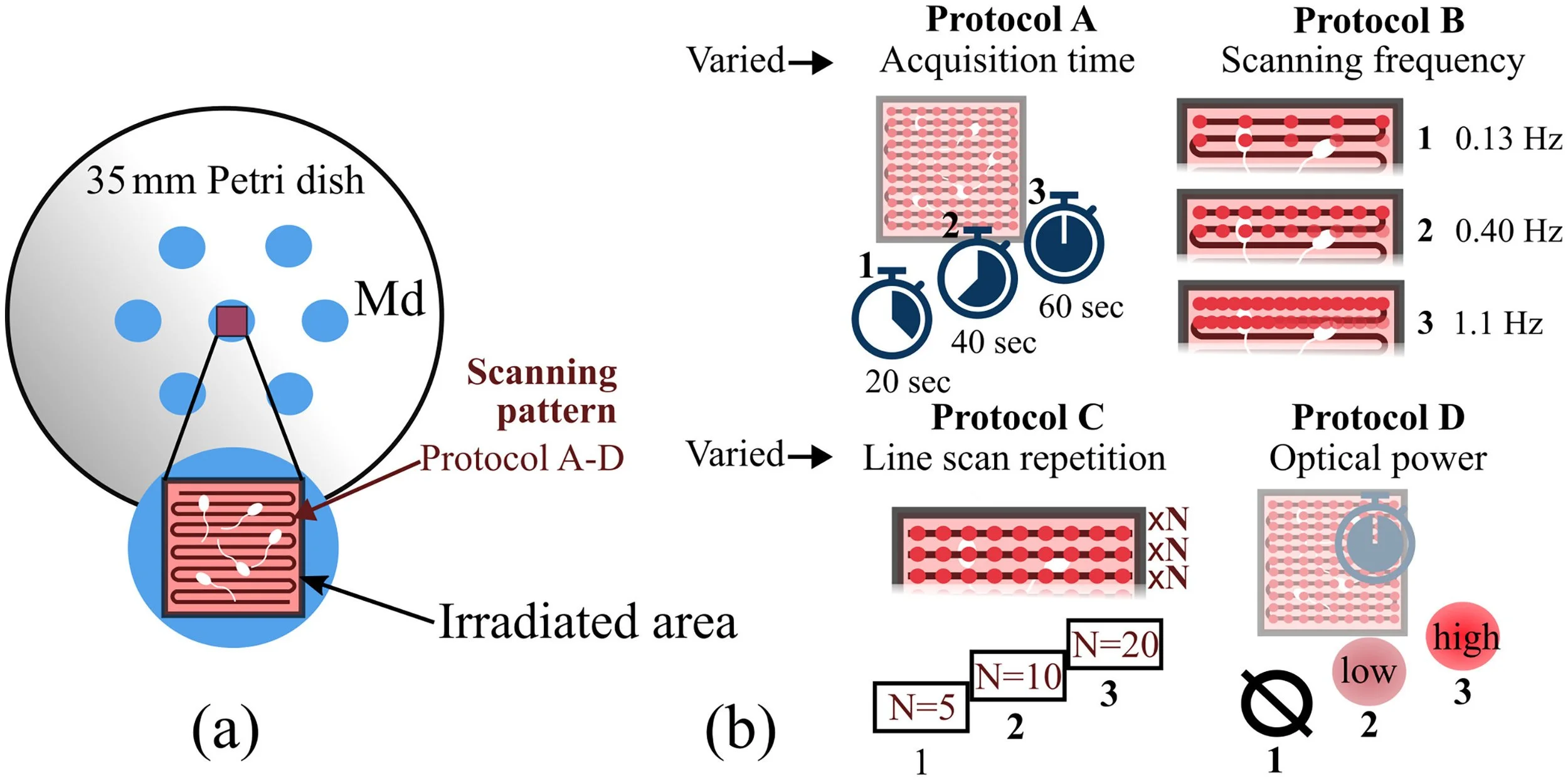
Boar semen irradiation protocolsThe irradiation protocols for the boar semen used the ‘irradiation mode’ configuration. The semen sample was prepared in a standard Petri dish, shown in Figure 2a. The irradiation followed three scanning protocols (A, B and C), which are described below and illustrated in Figure 2b. The mean ± SE optical power measured at the sample was 33 ± 3 mW at 1078 nm, which is 10–30 times more than the power value typically used for OCT imaging (Aumann et al., 2019).
Figure 2 (a) Diagram of Petri dish and arrangement of culture media droplets. The irradiated area is shown. (b) Description of the scanning protocols (A–D) showing the operation of the point-scanning optical coherence tomography system. Md, media droplet.
Protocols A and B simulated the conventional raster scanning pattern of an OCT system. Let X and Y be the axis of the horizontal plane, orthogonal to the optical axis, SMX scanned the beam rapidly along X, creating a line, and SMY scanned such line slowly along Y, together covering a square field of view. In Protocol A (Figure 2b), the volumetric scanning frequency (fV) was kept constant at 1.1 Hz, corresponding to a sampling density of 300 × 300 lateral points at 200 kHz, and the overall irradiation time (Ttotal) was varied from 20 to 60 s. In Protocol B (Figure 2b), Ttotal was kept constant at 60 s, and the scanning frequency was varied from 0.13 Hz to 1.1 Hz. Protocol C (Figure 2b) simulated a different OCT scanning pattern, termed ‘OCT angiography’ (OCT-A). In OCT-A, SMX scanned the same line a given number of times before SMY was moved to a neighbouring Y position.
Human semen irradiation protocolHuman semen samples were irradiated following Protocol D (Figure 2b). In Protocol D, both the scanning frequency and irradiation time were fixed (fV = 1.1 Hz, Ttotal = 60 s), and the irradiation power was varied. The semen samples were separated into three groups: low power, high power, and a non-irradiated control group. The low- and high-power conditions corresponded to an incident optical power on the sample of 4.9 ± 0.3 mW and 33 ± 3.0 mW, respectively, at a wavelength of 1078 nm. The power values were measured using a commercial power meter (PM100D; Thorlabs).
Statistical analysis for sperm parametersStatistical analysis, performed using SPSS version 28.0 (IBM, USA), was performed to evaluate whether irradiated samples (at different imaging protocols) showed significant differences from their matched manipulation control. The Kolmogorov–Smirnov test was used to assess normality, and groups were compared using the Kruskal–Wallis test. The number of degrees of freedom, H-values and P-values are given in tables for all comparisons. For all statistical comparisons, P-values <0.05 were considered to indicate significance.
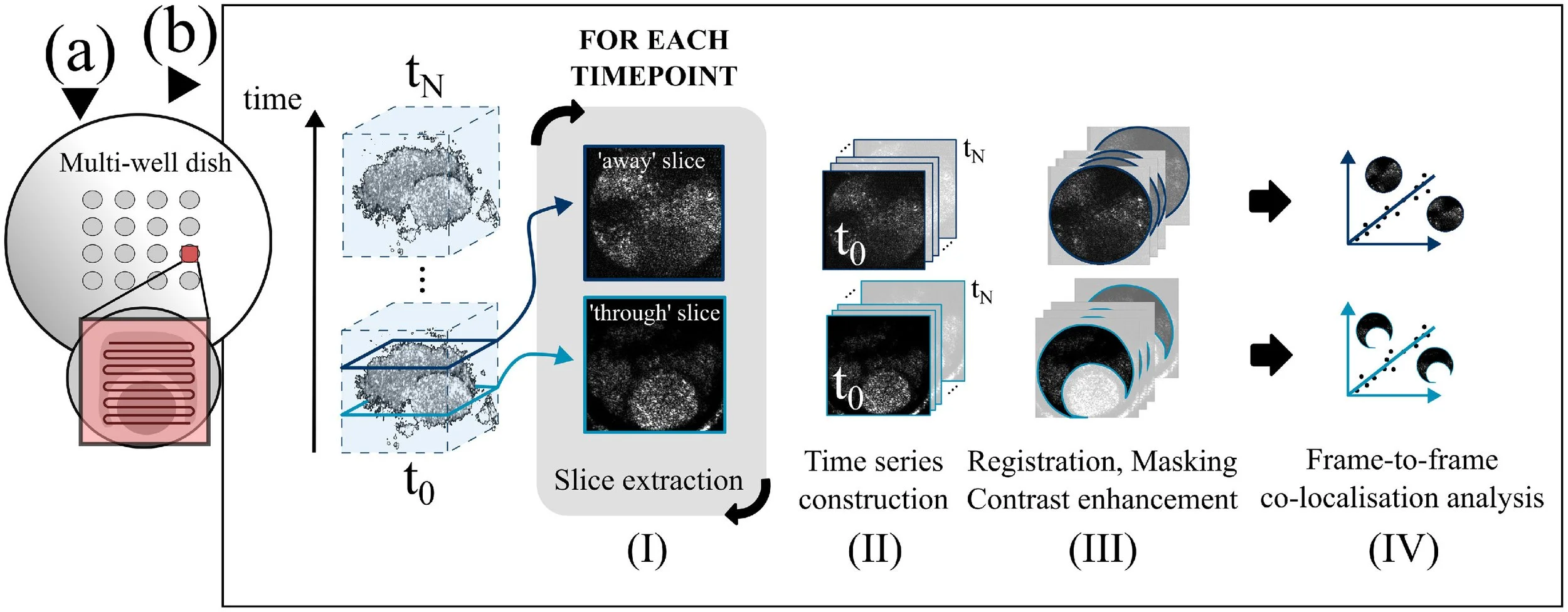
OCT imaging protocol for sperm incubation with cumulated oocytesFor OCT imaging of cumulus–oocyte complexes (COC) with added irradiated spermatozoa, porcine COC were retrieved from abattoir material as described in previous publications (Serrano Albal et al., 2022; Silvestri et al., 2020). This procedure yields mature oocytes surrounded by cumulus cells, but whose fertilization potential is highly dependent on the quality of the ovaries received from the abattoir. For the purposes of this study, they therefore serve as an indicator of sperm interaction with cumulus cells alone, not as a true readout of fertilization ability. Matured COC were pipetted into a multi-well Petri dish (Primo Vision, Gyncentrum, Poland), as shown in Figure 3a. The ‘OCT mode’ set-up (see Figure 1) was used to track porcine COC inside the incubator. Two wells, each containing a single COC, were imaged (n = 2): one with added irradiated boar spermatozoa (irradiated dish), and one without spermatozoa (control dish). The incident optical power with static beam was 1.3 mW at 1078 nm. OCT volumes of the irradiated dish were acquired every 5 min for 4 h, and OCT volumes of the control dish were acquired every 5 min for 3 h.
Figure 3 (a) Diagram of scanning pattern over sample in a well of a multi-well dish during the optical coherence tomography time lapse of porcine cumulus–oocyte complexes (COC) with irradiated spermatozoa. (b) Image processing routine for data analysis. For the three-dimensional dataset of each COC, co-localization analysis was performed at two depth slices: one intersecting the oocyte and part of the cumulus cells around the oocyte (‘through’ slice), and one above the oocyte, intersecting the cumulus cells alone (‘away’ slice).
The OCT stacks were processed with in-house LabVIEW software generating OCT images from raw OCT data, and an in-house Fiji (ImageJ) macro. The macro performs the steps of image stacking, image straightening, slicing, and construction of depth-specific time series. Frames from each time series are co-registered, then a mask is applied and the image contrast is enhanced (see Figure 3b). For each dataset, and for the processing steps presented hereafter, slicing was performed at two separate depths: one intersecting the oocyte and part of the cumulus cells around the oocyte (‘through’ slice), and another above the oocyte, intersecting the cumulus cells alone (‘away’ slice).
Co-localization analysisCo-localization methods aim to evaluate correlation between the pixel-wise intensity of two images. Here, a co-localization analysis was used to assess sample movement quantitatively in the cumulus cell area for both a control COC and a COC mixed with the irradiated sperm sample. The objective of this experiment was to compare the dynamics in the OCT image series between the two samples, and to determine whether it was possible to detect the cellular motions provoked by sperm interaction with, and penetration of, the cumulus cell mass. From the OCT time series, two depths were tracked in time for the irradiated dish and the control dish. The depths were chosen to include a portion of the cumulus cells: one intercepting the oocyte and masked as shown in Figure 3b, and one below the oocyte.
Co-localization analysis was performed using the JACoP plugin (Bolte and Cordelières, 2006). The Manders coefficient represents the fraction of the total intensity in the current image that co-localizes with the intensities in a reference image. Evolution of Manders coefficient M2 for each image between timepoints t1 and tn was evaluated, where the first image at timepoint t0 served as the reference image.
Logarithm intensity varianceThe co-localization analysis was complemented with visualization of the logarithm intensity variance (LIV). The LIV calculation measures the magnitude of the signal fluctuation on the total duration of the time lapse, highlighting movements in the sample (El Sadek et al., 2020). LIV processing was performed across 35 timepoints, acquired over a total time of 2 h and 50 min, for both the control COC and the COC mixed with the irradiated sperm sample. The algorithm used to build LIV mapping of the sample is adapted from Python software developed by El-Sadek et al. (2020).
Ethical approval and donor recruitmentThis study was approved by the Central Research Ethics Advisory Group at the University of Kent on 19 July 2022 (CREAG078-06-22), and adhered to the current legislation on research involving human subjects in the UK. Human volunteer donors (n = 6 samples analysed from three donors) were recruited via poster advertisement within the University of Kent.
Results
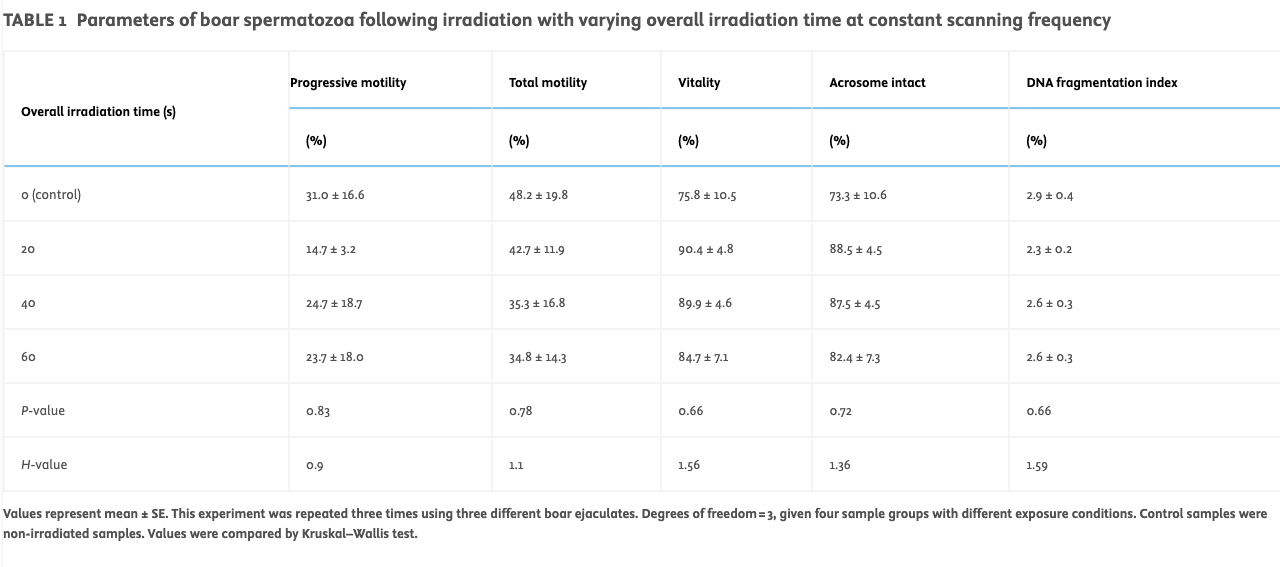
Varying irradiation time at constant scanning frequency and power (Protocol A)Exposure of boar spermatozoa (n = 3 ejaculates tested) in a commercial extender to varying overall exposure times (20, 40 and 60 s) resulted in no significant change in the tested sperm parameters (progressive motility, total motility, vitality, acrosome reactivity and sperm DNA damage; see Table 1).
TABLE 1
Parameters of boar spermatozoa following irradiation with varying overall irradiation time at constant scanning frequency
Values represent mean ± SE. This experiment was repeated three times using three different boar ejaculates. Degrees of freedom = 3, given four sample groups with different exposure conditions. Control samples were non-irradiated samples. Values were compared by Kruskal–Wallis test.
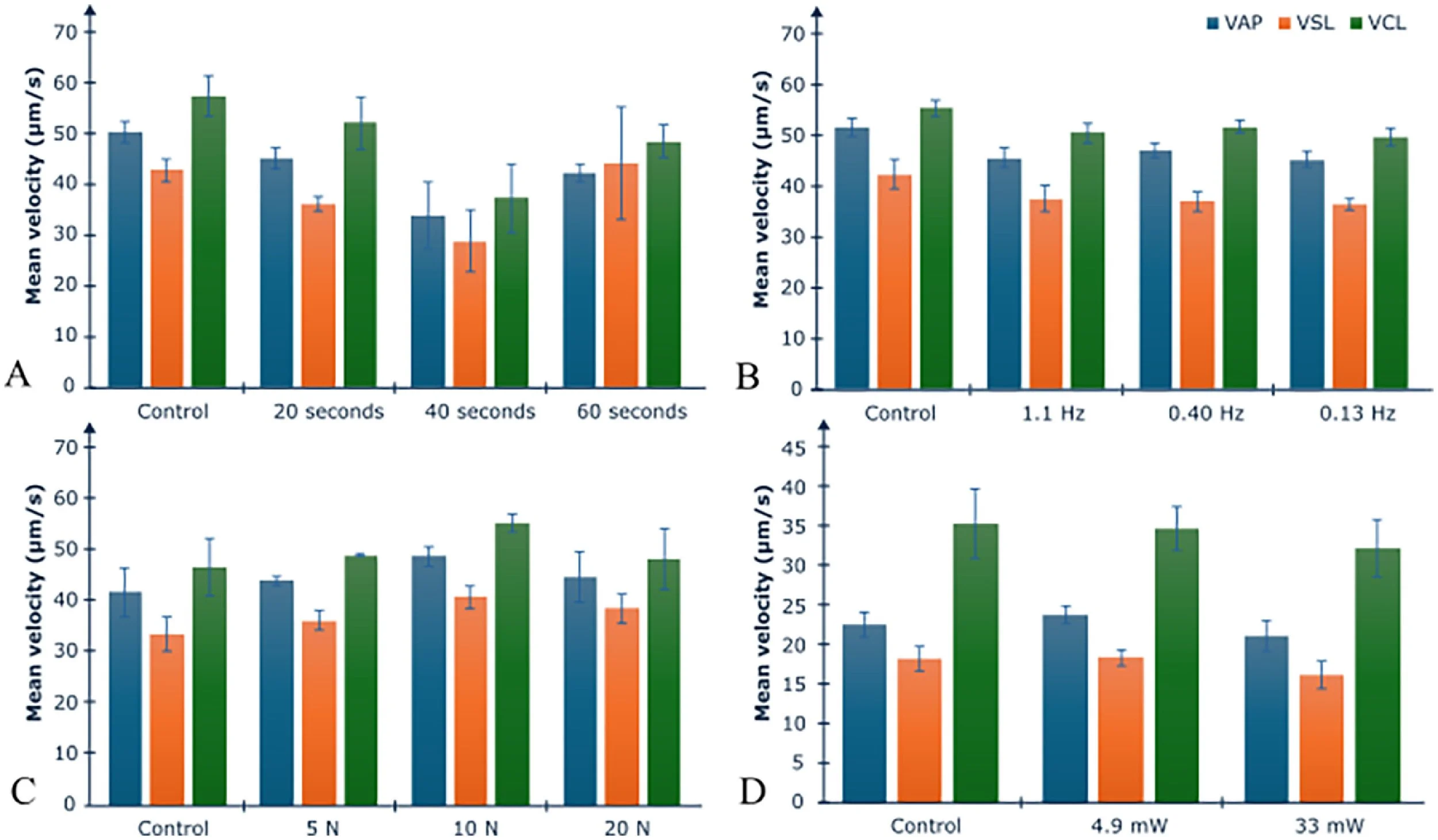
With regards to sperm kinetic (Figure 4A) parameters, the control showed no significant difference between VAP, VSL and VCL for the different irradiation times (20, 40 and 60 s).
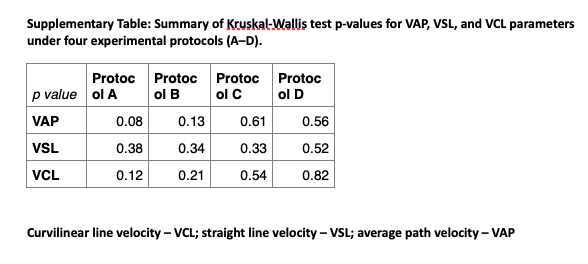
Figure 4 Boar and human sperm motility parameters when subjected to various conditions (Protocols A–D): (A) boar spermatozoa subjected to varied overall irradiation time; (B) boar spermatozoa subjected to varied scanning frequency; (C) boar spermatozoa subjected to varying optical coherence tomography angiography line scan repetition; (D) human spermatozoa irradiated with two different optical powers. Control samples are non-irradiated samples. No significant differences were found between groups for average path velocity (VAP), straight line velocity (VSL) and curvilinear line velocity (VCL) for each protocol according to the Kruskal–Wallis test (see Supplementary Table for P-values). N, number of scan repetitions. Error bars represent standard error.
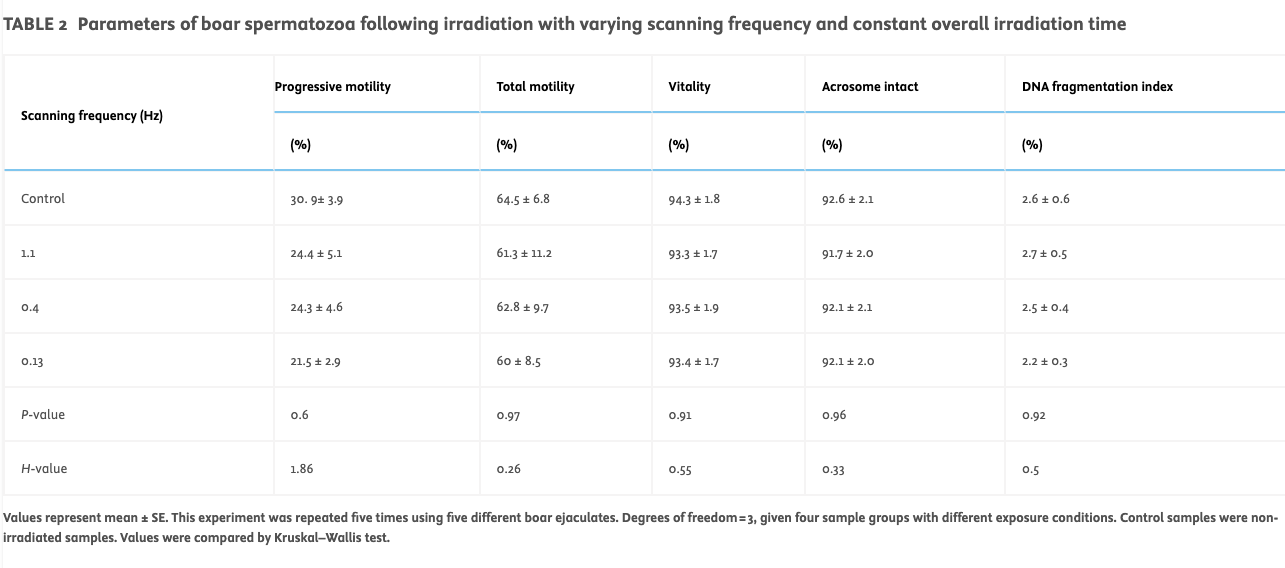
Varying scanning frequency at constant irradiation time and power (Protocol B)Exposure of boar spermatozoa (n = 5 ejaculates tested) in a commercial extender to varying scanning frequencies, at an irradiation time of 60 s, resulted in no significant change in the tested sperm parameters (progressive motility, total motility, vitality, acrosome reactivity and sperm DNA damage; see Table 2).
TABLE 2
Parameters of boar spermatozoa following irradiation with varying scanning frequency and constant overall irradiation time
Values represent mean ± SE. This experiment was repeated five times using five different boar ejaculates. Degrees of freedom = 3, given four sample groups with different exposure conditions. Control samples were non-irradiated samples. Values were compared by Kruskal–Wallis test.
With regards to sperm kinetic (Figure 4B) parameters, the control showed no significant difference between VAP, VSL and VCL for the varying scanning frequencies.
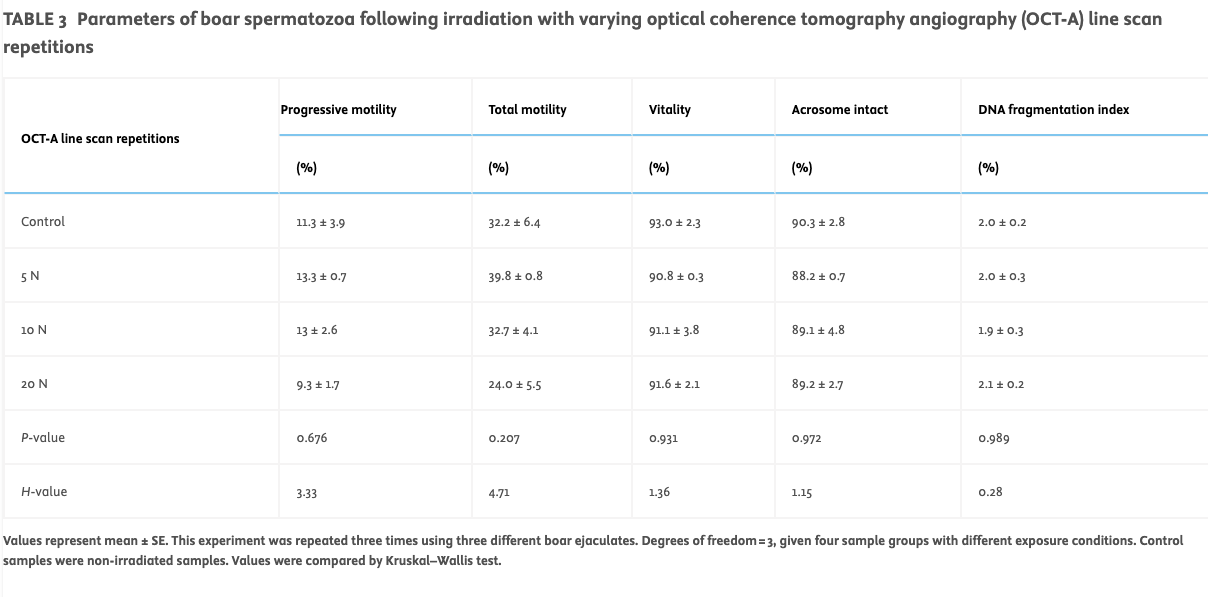
Varying OCT-A line scan repetition rate at constant irradiation time, scanning frequency and power (Protocol C)Exposure of boar spermatozoa (n = 3 ejaculates tested) in a commercial extender to various numbers of OCT-A line scan repetitions (five, 10 and 20) resulted in no significant change in the tested sperm parameters (progressive motility, total motility, vitality, acrosome reactivity and sperm DNA damage; see Table 3).
TABLE 3
Parameters of boar spermatozoa following irradiation with varying optical coherence tomography angiography (OCT-A) line scan repetitions
Values represent mean ± SE. This experiment was repeated three times using three different boar ejaculates. Degrees of freedom = 3, given four sample groups with different exposure conditions. Control samples were non-irradiated samples. Values were compared by Kruskal–Wallis test.
With regards to sperm kinetic (Figure 4C) parameters, the control showed no significant difference between VAP, VSL and VCL for the varying number of OCT-A line scan repetitions.
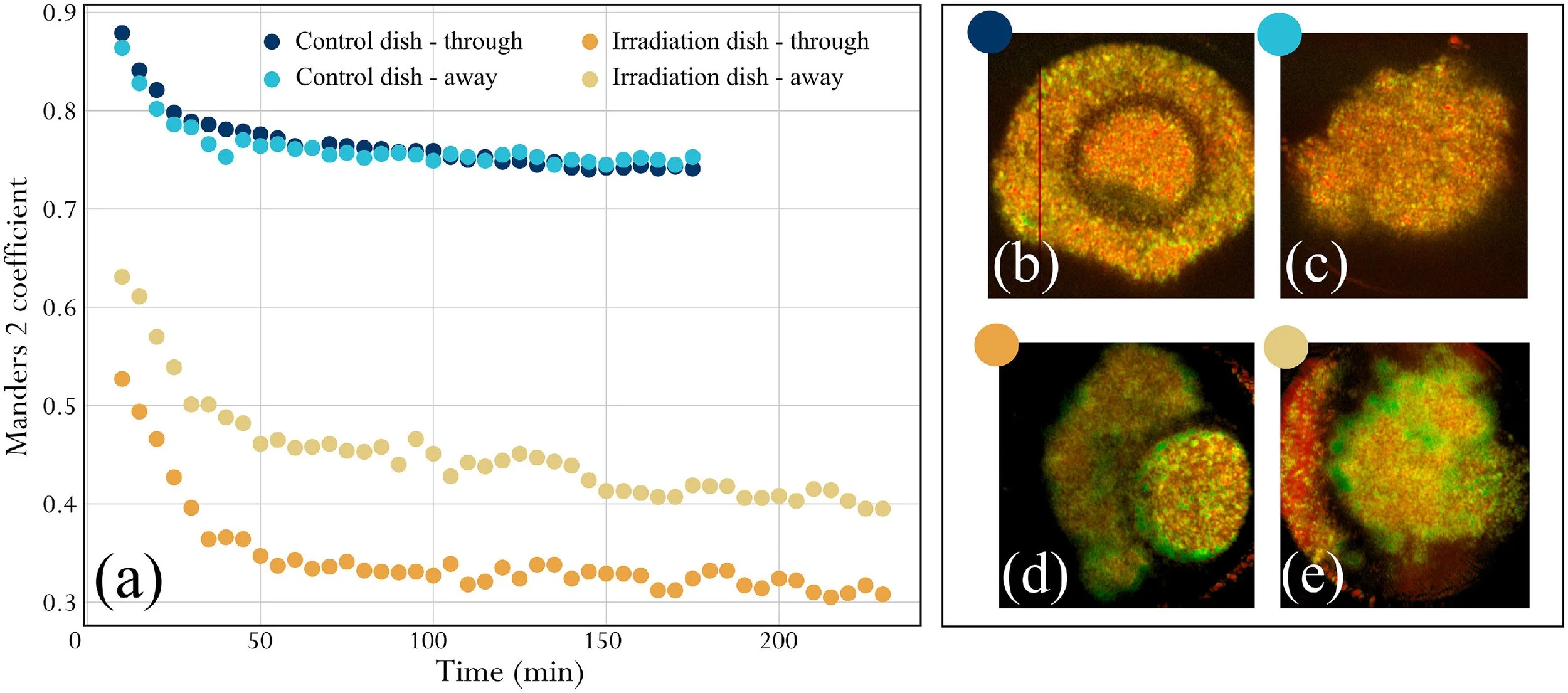
Boar spermatozoa remain competent to interact with and enter the cumulus following OCT irradiationHaving demonstrated that none of the above exposure parameters altered sperm motility, acrosome status or DNA fragmentation, even at values higher than those typically used for OCT imaging, a pilot study was conducted to test sperm behaviour in co-culture with COC to determine whether this interaction was perturbed by OCT imaging. The ‘OCT mode’ set-up was employed to monitor the in-situ interaction of spermatozoa with COC (n = 2 COC tested) within the incubator environment (see Supplementary Video). The first COC was co-incubated with irradiated boar spermatozoa (irradiated dish), while the second COC served as a control (i.e. without the addition of spermatozoa) (control dish). Figure 5a shows the result of the co-localization analysis (M2 coefficient) for both depths in the irradiated dish and the control dish. For the control dish, the M2 value is high for the first frames, and decreases slowly throughout the time series for the control dish while staying above 0.7, indicating very small frame-to-frame variation (i.e. slow dynamics). For the irradiated dish, plotting of the M2 value showed a linear decrease between frames 1 and 6 (30-min duration), and a second linear decrease with a less steep slope for the rest of the time series, down to M2 values between 0.3 and 0.4. These results highlight an additional dynamic activity in the irradiated dish compared with the control dish, likely resulting from sperm movement within the cumulus cells, and suggesting a fair level of sperm mobility.
Figure 5 Analysis of sample dynamics across the time-lapse duration. (a) Co-localization analysis, evolution of M2 coefficient between a given frame and the reference frame at t0. The evolution of M2 against time is shown in shades of blue for the control cumulus–oocyte complex (COC) (control dish), and in shades of yellow for the irradiated COC (irradiation dish). For each sample, data from two depths are considered. Frames were acquired at 5-min intervals. (b–e) Logarithm intensity variance (LIV) maps obtained with the same frames. In LIV maps, red corresponds to low dynamics and green corresponds to high dynamics. Map brightness corresponds to the logarithmic optical coherence tomography intensity.
To validate the co-localization analysis, LIV maps of the irradiated dish and control dish at the same depths of interest were computed and are presented in Figure 5b–e. LIV maps corresponding to the irradiated dish (Figure 5d,e) showed more green areas in the cumulus cells compared with LIV maps corresponding to the control dish (Figure 5b,c). This indicates much higher dynamic activity in the irradiated dish, further validating the conclusions drawn from the co-localization analysis. Of note, the high LIV signal in Figure 5d,e is present throughout the cumulus mass, and is not confined to the surface layer, indicating that the spermatozoa are fully penetrating the cumulus during this experiment.
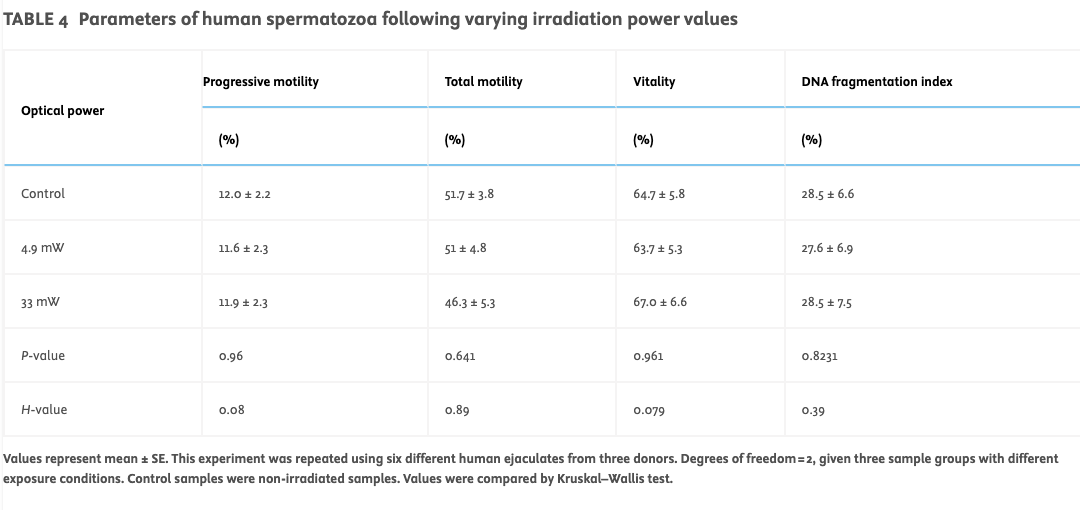
Human spermatozoa show no adverse effects of OCT illumination even at powers exceeding typical imaging (Protocol D)As boar sperm chromatin is typically more resistant to damage than human sperm chromatin, the effect of irradiation of human semen (n = 6 ejaculates tested from three donors) was tested at different power levels using the harshest set of parameters tested previously on boar spermatozoa (Protocol D). For this analysis, laser power levels substantially exceeding those typically used for OCT imaging were used to determine whether this provoked any detectable change in semen parameters.
Exposure of human semen samples at a scanning frequency of 1.10 Hz for 60 s at varied power (4.9 ± 0.3 mW and 33 ± 3 mW at 1078 nm) resulted in no significant change in the tested parameters across the different groups (see Table 4). With regards to sperm kinetic (Figure 4D) parameters, the control showed no significant difference in VAP, VSL and VCL for the two irradiation powers tested.
TABLE 4
Parameters of human spermatozoa following varying irradiation power values
Values represent mean ± SE. This experiment was repeated using six different human ejaculates from three donors. Degrees of freedom = 2, given three sample groups with different exposure conditions. Control samples were non-irradiated samples. Values were compared by Kruskal–Wallis test.
Discussion
While multiple studies have focused on the potential in-vitro and in-vivo use of OCT for imaging during embryogenesis, few have explored the safety of OCT in relation to genotoxic effects, and none have studied its effects on spermatozoa. Therefore, to the authors’ knowledge, this is the first systematic study to investigate the potential genotoxic impact of broadband infra-red radiation on boar and human spermatozoa. This study found no observable effect on sperm parameters following exposure to broadband infra-red irradiation, and found that OCT can detect the motion of cumulus cells caused by interactions with live sperm cells. Thus, should future tests also confirm its safety for oocytes and zygotes, OCT could, in principle, be used as a safe method to enable 3D morphokinetic investigation of the very earliest events in mammalian life, even prior to and during fertilization.
In particular, this study showed that boar spermatozoa exposed to broadband infra-red light around 1060 nm at varying exposure times and scanning frequencies showed no changes in vitality, motility patterns, acrosome integrity or DNA fragmentation, even when using power levels 10–30 times higher than commonly employed in OCT imaging. This result was replicated for human spermatozoa, and even at the highest power level of 33 mW, there was no effect on sperm parameters, including DNA damage levels. It is also worth noting that while the boar samples used in this study had very low baseline DNA fragmentation of 2–3% (typical for stud animals highly selected for fertility), the volunteer donor human semen samples used in this study had moderate baseline DFI levels of approximately 30%. This is at the high end of normal DFI for unselected humans, likely reflecting the fact that the donors were not required to observe the strict rules on ejaculation frequency typically used in diagnostic semen analysis. Therefore, the lack of a detectable change in DNA fragmentation in response to OCT illumination is a genuine finding, and is not due to any lack of sensitivity in the DFI detection protocol.
In addition, this study found no difference in acrosome reactivity or motility in the irradiated samples. The acrosome reaction is a critical process for successful fertilization (Hirohashi and Yanagimachi, 2018; Tello-Mora et al., 2018). The finding that infra-red illumination does not impact the proportion of boar spermatozoa undergoing the acrosome reaction suggests that the irradiation does not compromise the structural or functional integrity of the sperm membrane, confirming the preserved fertilization potential of spermatozoa. Interestingly, some studies have shown improved sperm motility and calcium transport, as well as no significant change in sperm DNA damage levels following low-power monochromatic visible irradiation at 633 nm (Breitbart et al., 1996; Preece et al., 2017) and at 905 nm (Firestone et al., 2013). However, the present study found no difference in sperm motility following infra-red illumination, likely due to the different irradiation protocols followed and different wavelengths.
How could OCT be deployed within the clinical environment? There are several areas where it may be useful. In particular, the high-resolution depth sectioning ability of OCT imaging has the potential to provide additional information for selecting the optimal embryo for elective transfer, mitigating risks linked to multi-embryo transfer such as multiple pregnancy. The incorporation of OCT technology into traditional time-lapse systems could also allow for the identification of potential embryonic defects that might not be observable through conventional analysis methods. Most pertinent to this study on gamete safety, OCT could potentially be used for guiding the positioning of micro-injection needles during the intracytoplasmic sperm injection procedure, helping to avoid damage to the meiotic spindle. Finally, as demonstrated in this study, movements of cumulus cells during co-incubation with spermatozoa can be observed – this could potentially be used to track the progress of cumulus penetration, and help diagnose patients with fertility problems affecting this process.
Limitations of the study and directions for future workThe primary limitation of this study is the low number of samples available for the pilot data on cumulus penetration (n = 2 COC) and human donor studies (n = 6 from three donors). Thus, the authors do not claim that these findings are sufficient to support the clinical use of OCT at this stage. Rather than extrapolating safety conclusions directly to clinical practice, the aim of this study was to establish a foundation for assessing potential DNA or functional damage using sensitive and relevant biological readouts. These pilot data provide preliminary reassurance that the tested OCT parameters do not induce overt harm under the experimental conditions used, but it is fully acknowledged that broader validation is essential.
A further limitation is that acrosome integrity was only assessed in boar spermatozoa and not in human spermatozoa. This will be assessed in future studies. Finally, while this work provides encouraging preliminary data to suggest that acute OCT illumination has no adverse effects on human semen samples, a wider study including donors with varying levels of baseline sperm parameters will be needed to establish its utility in patient populations. For longer term OCT monitoring of embryo development, further research will also need to address the potential effects of cumulative/long-term OCT exposure.
While this study serves as a proof of concept to show that spermatozoa retain the ability to penetrate the cumulus following irradiation, and that OCT can detect the movements caused by this, further experimentation will be required to understand how robust this is to the experimental set-up, and how defects in different stages of fertilization affect the motions visible by OCT. In particular, mouse models with defects in cumulus penetration (e.g. deficiencies in hyaluronidase or acrosomal proteases) (Kawano et al., 2010; Park et al., 2019) versus zona binding (reviewed by Inoue et al., 2007) versus oolemma binding (e.g. Izumo1 deficiency) (Matsumura et al., 2022) could reveal how sensitive OCT imaging is to different stages of the fertilization process, and whether this could be useful in defining different groups of infertile patients.
In terms of observing the moment of fertilization itself, although OCT images were obtained during cumulus penetration, the authors were not able to observe fertilization definitively due to difficulties obtaining viable oocytes from abattoir material. Thus, the results from this feasibility study warrant further investigation into analysing the fertilization and embryo development potential following gamete and embryo irradiation to develop a more comprehensive embryo imaging technology. Additional testing will involve the use of an integrated time-lapse system utilizing OCT technology to optimize embryo monitoring and selection throughout the entire early embryonic development.
Extensive testing in animal model systems will be required before contemplating work on human embryos, including demonstration of successful fertilization and early embryo development. Given typical success rates from fertilization through to blastocyst formation of approximately 80% in murine IVF (Vasudevan et al., 2010) and approximately 30% in porcine IVF (Chen et al. 2022), a cohort size of approximately 1100 embryos (mouse) or 1250 embryos (pig) per experimental group would be required to detect a 5 percentage point change in blastulation rate. Following this, further work should involve fertilization and embryo development endpoints in appropriate preclinical models. If safety is confirmed in these expanded studies, potential clinical applications – such as real-time sperm selection or diagnostic imaging – could be considered. Until then, the authors view these findings as an initial step towards defining the experimental and safety framework required for such translation.
Acknowledgments
Author contributionsConception and design of the study: GS, MJM, JC, MCA, PJE. Experimental work and data acquisition: WI, TS, LA, MCA, JC, MJM. Analysis and interpretation of data: MCA, JC, PJE, GS, MJM, WI, TS, LA, AP, DG. Drafting the article or revising it critically for important intellectual content: MCA, JC, PJE, MM. All authors read and approved the final manuscript.
FundingThis research was supported by the Summer Vacation Research Competition 2022, Graduate Researcher College, University of Kent. JC, MJM, AP and DG acknowledge BBSRC for research grant 5DHiResE (BB/S016643/1). GS, PE and AP wish to thank the MRC, Impact Accelerator Account (Project 18557). In addition, the authors acknowledge Superlum Diodes for the loan of the MOPA source prototype.
Appendix Supplementary Materials (3)
Supplementary Video: 4-hour OCT timelapse of a single selected depth slice, for the Irradiation dish (left) and for the Control dish (right). In both timelapse videos the chosen depth slice intersects the oocyte and surrounding cumulus cells (‘through’ slice, as defined in Methods).
-
Alam Z, Poddar R.
An in-vivo depth-resolved imaging of developing zebrafish microstructure and microvasculature using swept-source optical coherence tomography angiography.
Optics and Lasers in Engineering. 2022;156:107087.Apelian C, Harms F, Thouvenin O, et al.
Dynamic full field optical coherence tomography: subcellular metabolic contrast revealed in tissues by interferometric signals temporal analysis.
Biomedical Optics Express. 2016;7:1511–1524.Aumann S, Donner S, Fischer J, et al.
Optical Coherence Tomography (OCT): Principle and Technical Realization.
In: Bille JF, ed. High Resolution Imaging in Microscopy and Ophthalmology. Springer; 2019:59–85.Bahat A, Eisenbach M.
Human sperm thermotaxis is mediated by phospholipase C and inositol trisphosphate receptor Ca²⁺ channel.
Biology of Reproduction. 2010;82:606–616.Bolte S, Cordelières FP.
A guided tour into subcellular colocalization analysis in light microscopy.
Journal of Microscopy. 2006;224:213–232.Breitbart H, Levinshal T, Cohen N, et al.
Changes in calcium transport in mammalian sperm mitochondria and plasma membrane irradiated at 633 nm (HeNe laser).
Journal of Photochemistry and Photobiology B. 1996;34:117–121.Castellò D, Motato Y, Basile N, et al.
How much have we learned from time-lapse in clinical IVF?
Molecular Human Reproduction. 2016;22:719–727.Cernat R, Tatla TS, Pang J, et al.
Dual instrument for in vivo and ex vivo OCT imaging in an ENT department.
Biomedical Optics Express. 2012;3:3346–3356.Chamayou S, Patrizio P, Storaci G, et al.
The use of morphokinetic parameters to select all embryos with full capacity to implant.
Journal of Assisted Reproduction and Genetics. 2013;30:703–710.Chen PR, Uh K, Redel BK, et al.
Production of pigs from porcine embryos generated in vitro.
Frontiers in Animal Science. 2022;3:826324.Cruz M, Garrido N, Herrero J, et al.
Timing of cell division in human cleavage-stage embryos is linked with blastocyst formation and quality.
Reproductive BioMedicine Online. 2012;25:371–381.Dal Canto M, Coticchio G, Mignini Renzini M, et al.
Cleavage kinetics analysis of human embryos predicts development to blastocyst and implantation.
Reproductive BioMedicine Online. 2012;25:474–480.El-Sadek I, Miyazawa A, Shen LTW, et al.
Optical coherence tomography-based tissue dynamics imaging for tumor spheroids.
Biomedical Optics Express. 2020;11:6231–6248.Enciso M, Johnston SD, Gosálvez J.
Differential resistance of mammalian sperm chromatin to oxidative stress.
Reproduction, Fertility and Development. 2011;23:633–637.Evenson DP.
Sperm Chromatin Structure Assay (SCSA®).
In: Carrell DT, Aston KI, eds. Spermatogenesis: Methods and Protocols. Humana Press; 2013:147–164.Firestone RS, Esfandiari N, Moskovtsev SI, et al.
Effects of low-level laser light exposure on sperm motion and DNA damage.
Journal of Andrology. 2013;33:469–473.Fluks M, Tamborski S, Szkulmowski M, et al.
Optical coherence microscopy for immature mouse oocyte assessment.
Reproduction. 2022;164:83–95.García-Rodríguez A, Gosálvez J, Agarwal A.
DNA damage and repair in human reproductive cells.
International Journal of Molecular Sciences. 2018;20:31.González-Marín C, Gosálvez J, Roy R.
Types and detection of DNA fragmentation in sperm cells.
International Journal of Molecular Sciences. 2012;13:14026–14052.Hirohashi N, Yanagimachi R.
Sperm acrosome reaction: site and role.
Biology of Reproduction. 2018;99:127–133.Holmes J.
In vivo real-time OCT imaging of Drosophila.
Nature Methods. 2009;6:10.Huang D, Swanson EA, Lin CP, et al.
Optical coherence tomography.
Science. 1991;254:1178–1181.Inoue N, Yamaguchi R, Ikawa M.
Sperm-egg interaction and gene-manipulated animals.
Society of Reproduction and Fertility Supplement. 2007;65:363–371.Karnowski K, Ajduk A, Wieloch B, et al.
Optical coherence microscopy for 4D live embryo imaging.
Scientific Reports. 2017;7:4165.Kawano N, Kang W, Yamashita M, et al.
Sperm serine proteases ACR and PRSS21 deficiency.
Biology of Reproduction. 2010;83:359–369.Khodavirdilou R, Pournaghi M, Oghbaei H, et al.
Toxic effect of light on oocytes and embryos.
Archives of Toxicology. 2021;95:3161–3169.Kirkegaard K, Hindkjaer JJ, Grøndahl ML, et al.
Embryo culture: conventional vs time-lapse incubator.
Journal of Assisted Reproduction and Genetics. 2012;29:565–572.Kohlfaerber T, Pieper M, Münter M, et al.
Dynamic microscopic OCT of airways.
Biomedical Optics Express. 2022;13:3211–3223.Kovacs P.
Embryo selection and time-lapse monitoring.
Reproductive Biology and Endocrinology. 2014;12:124.Masuda Y, Hasebe R, Kuromi Y, et al.
Hatchability evaluation of bovine IVF embryos using OCT-based 3D analysis.
Journal of Reproduction and Development. 2023;69:239–245.Matsumura T, Noda T, Satouh Y, et al.
Sperm IZUMO1 requirement for binding preceding fusion.
Frontiers in Cell and Developmental Biology. 2022;9:810118.Ménézo Y, Dale B, Cohen M.
DNA damage and repair in human oocytes and embryos.
Zygote. 2010;18:357–365.Mortimer ST.
CASA – practical aspects.
Journal of Andrology. 2000;21:515–524.Nakahara T, Iwase A, Goto M, et al.
Safety of time-lapse observations for embryos.
Journal of Assisted Reproduction and Genetics. 2010;27:93–96.Ottosen LD, Hindkjaer J, Ingerslev J.
Light exposure during ART procedures.
Journal of Assisted Reproduction and Genetics. 2007;24:99–103.Park S, Kim YH, Jeong PS, et al.
SPAM1/HYAL5 double deficiency and male subfertility.
FASEB Journal. 2019;33:14440–14449.Petersen BM, Boel M, Montag M, et al.
Morphokinetic algorithm predicting implantation potential.
Human Reproduction. 2016;31:2231–2244.Popescu DP, Choo-Smith LP, Flueraru C, et al.
Optical coherence tomography: principles and applications.
Biophysical Reviews. 2011;3:155.Preece D, Chow KW, Gomez-Godinez V, et al.
Red light improves sperm motility without DNA damage.
Scientific Reports. 2017;7:46480.Ren C, Hao S, Wang F, et al.
Dynamic contrast optical coherence tomography (DyC-OCT).
Communications Biology. 2024;7:278.Robles V, Martínez-Pastor F.
Flow cytometric methods for sperm assessment.
Methods in Molecular Biology. 2013;927:175–186.Rodríguez-Gil JE.
Photostimulation and thermotaxis of sperm.
Theriogenology. 2019;137:8–14.Serrano Albal M, Silvestri G, Kiazim LG, et al.
FGF2, LIF, IGF1 supplementation in porcine IVM.
Zygote. 2022;30:801–808.Sepehr A, Armstrong WB, Guo S, et al.
Optical coherence tomography of the larynx.
Otolaryngology–Head and Neck Surgery. 2008;138:425–429.Silvestri G, Rathje CC, Harvey SC, et al.
Optimal assisted aspiration conditions of oocytes.
Veterinary Medicine and Science. 2020;7:465–473.Storr A, Venetis C, Cooke S, et al.
Time-lapse algorithms vs morphological selection.
Fertility and Sterility. 2018;109:276–283.e3.Takae S, Tsukada K, Sato Y, et al.
Ovarian reserve assessment using OCT.
Scientific Reports. 2017;7:43550.Tello-Mora P, Hernández-Cadena L, Pedraza J, et al.
Acrosome reaction and chromatin integrity.
Reproductive Biology and Endocrinology. 2018;16:102.Vasilyeva TA, Marakhonov AV, Sukhanova NV, et al.
Preferentially paternal origin of de novo 11p13 deletions.
Genes. 2020;11:812.Vasudevan K, Raber J, Sztein J.
Fertility comparison between wild type and transgenic mice.
Transgenic Research. 2010;19:587–594.World Health Organization.
WHO Laboratory Manual for the Examination and Processing of Human Semen. 5th ed. Geneva; 2010.Wyck S, Herrera C, Requena CE, et al.
Oxidative stress in sperm affects epigenetic reprogramming.
Epigenetics & Chromatin. 2018;11:60.
Key message
Broadband infra-red radiation at the intensities typically used for optical coherence tomography has no effect on boar or human sperm parameters, and irradiated boar spermatozoa retain the ability to penetrate the cumulus oophorus. This provides a step towards development of non-invasive methods to monitor early embryonic events in three dimensions.