Environmental Validation of a Novel Automated IVF Platform: Results from Comprehensive Off-Gassing and Toxicity Assessment
Flores-Saiffe Farias et al., American Society for Reproductive Medicine (ASRM) 2025 Scientific Congress & Expo
Objective
To establish whether AURA, an automated IVF platform, meets environmental safety standards for embryo development through air quality, embryotoxicity, and microbiological testing.
Materials and Methods
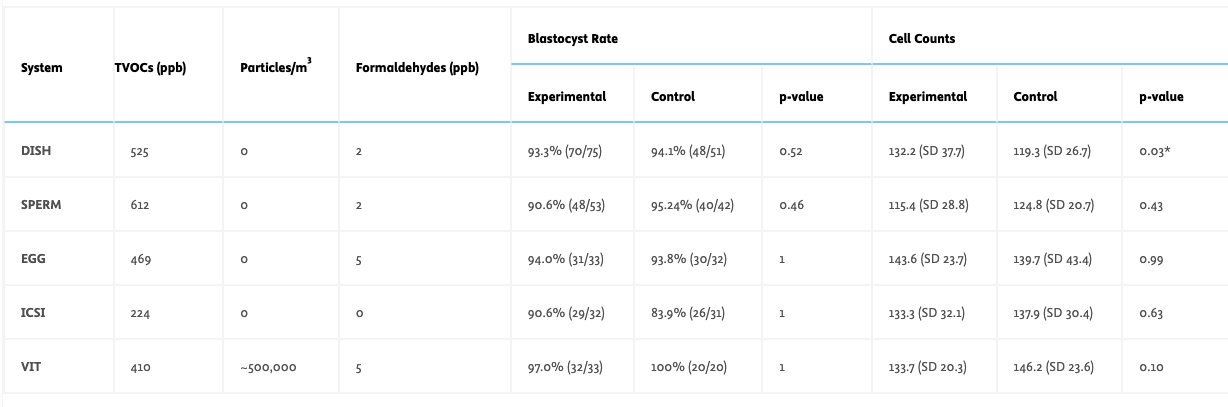
AURA comprises five subsystems: dish preparation (C:DISH), sperm preparation (C:SPERM), oocyte preparation (C:EGG), fertilization (C:ICSI), and vitrification (C:VIT). All subsystems underwent off-gassing (35-40°C, 3-15 days) with daily measurements of TVOCs, formaldehyde (GrayWolf PCC-20A), and particles (≥0.3 μm, Extech VPC260). Mouse Embryo Assays (MEAs) evaluated embryotoxicity using B6/CBA zygotes cultured in media exposed to subsystem environment overnight. Blastocyst formation and day-6 cell counts were analyzed (Fisher's Exact Test; Mann-Whitney U Test). Controls included laboratory air and unexposed embryos for MEAs. Microbiological testing verified Grade A cleanroom compliance (<1 CFU/m3 air; <1 CFU/plate surface).
Results
Off-gassing achieved stable TVOC concentrations (400-500 ppb) comparable to reference IVF laboratories. Particle counts reached zero in four subsystems. Formaldehyde decreased below 5 ppb in all components, meeting Cairo consensus 2018 standards. MEAs showed high two-cell rates (>95%) and blastocyst formation (>80%) with no significant differences between experimental and control groups. Good quality blastocyst rates >50% with cell counts >110 were observed in both groups. No significant differences in cell counts were found (p>0.05), except C:DISH showed higher counts than controls (p=0.03). Microbiological sampling confirmed Grade A standards compliance.
Conclusions
Environmental validation confirms AURA's suitability for gamete and embryo manipulation. Integrated off-gassing, air monitoring, MEAs, and microbiological surveillance provide a framework for validating biosafety, paving the way for clinical evaluation.
Impact Statement
This work establishes a multi-parameter approach for environmental validation of automated IVF equipment, demonstrating that robotic systems can achieve high environmental standards comparable to traditional IVF laboratories.
Support
Financial Support: Institutional