Activity-Based Costing in IVF: A Framework for Transparency and Operational Scaling of Fertility Services
Chavez-Badiola et al., Journal of Assisted Reproduction and Genetics (JARG), 2025
Table Of ContentsIntroduction
Reframing IVF Labs
Activity-Based Costing
Multi-Clinic Analysis
Data Collection/Validation
Labor Economies/Queueing
Cost Structure Modeling
Cost Elasticity Calculations
Scale Economics
ABC Applications
Capacity Planning
Capital Investment Consolidation Economics
Business Model Innovation
Implementation Challenges
Ethical Implementation
Conclusions
Data Availability
Supplementary Information
Abstract
PurposeThe economics of IVF operations remain opaque despite growing prevalence and substantial investment in fertility services. Traditional accounting methods often fall short by failing to capture the complex relationship between biological inputs (number of eggs and embryos processed), skilled labor, and equipment utilization, creating a distorted view across different procedures and patient characteristics.
MethodsThis paper presents Activity-Based Costing (ABC) as a methodological framework for understanding true IVF laboratory costs. We demonstrate a use case with an analysis of data from five IVF laboratories (600–2200 annual egg retrieval cycles) across different US regions including time studies of 500 + procedures at each site, with theoretical projections to 4000 annual egg retrieval cycles based on queuing theory.
ResultsWe identify significant cost elasticity with biological inputs; costs increase by 55% for egg freezing when the number of eggs per patient triples, and by 30% for complex IVF procedures (such as those requiring embryo biopsy) when egg count per patient doubles.
We also describe substantial scale effects, with marginal lab costs 40% higher in centers performing 500 yearly egg retrieval cycles versus 2000-cycle centers, and 4000-cycle centers estimated to achieve 13–40% lower costs per procedure than operations performing 500 retrieval cycles annually. Finally, we show labor efficiency increases by 30% between centers performing 500 and 2000 annual egg retrieval cycles, an observation supported by a queuing rule of thumb (QROT) analysis, with capital utilization also improving dramatically.
ConclusionsABC enables evidence-based pricing, capacity planning, technology assessment, and consolidation strategizing. ABC is intended to inform operational efficiency, not clinical treatment, ensuring that quality of care and patient outcomes remain the primary considerations in fertility services.
KEY WORDSAssisted Reproductive Techniques • Fertility • Infertility • Polycystic ovary syndrome • Reproductive Medicine • Reproductive Physiology
Introduction
The IVF field stands at a critical inflection point. Growing demand, consolidation pressure, and technological advancement create a complex operational landscape requiring sophisticated financial tools [1]. Despite rapid market growth, the economics of IVF operations remain poorly understood, with most clinics relying on simplistic accounting methods that fail to capture the complex interplay between biological inputs, labor requirements, and fixed infrastructure costs.
This opacity affects multiple dimensions of the fertility sector:
Pricing transparency: Patients face widely varying prices with little understanding of underlying cost drivers.
Investment decisions: Clinic owners lack robust frameworks for evaluating technology and facility investments.
Operational planning: Laboratory directors, CFOs, and clinical operators struggle to optimize workflows, scheduling, and staffing without granular cost data, particularly in consolidated networks where decision-making authority may be distributed across multiple roles.
Consolidation strategies: Acquiring organizations cannot accurately quantify scale benefits or synergies.
Reframing IVF Labs as Specialized Manufacturing Operations
To properly understand IVF laboratory economics, we must first reconceptualize these facilities as what they truly are: specialized operations that convert inputs (eggs, sperm, laboratory consumables, labor) into outputs (embryos, pregnancies) through a complex series of activities requiring specialized equipment and environmental conditions which, to a certain extent, must be adapted to the individual case.
Traditional healthcare accounting focused on simple metrics like “cost per cycle” fails to capture this complexity. A manufacturing lens reveals the critical distinction between:
Fixed costs: Setup, documentation, witnessing, and cleanup activities that remain relatively constant regardless of biological volume.
Variable costs: Processing time, consumables, and monitoring requirements that scale with egg or embryo count.
Equipment utilization: Specialized capital assets with usage patterns that vary dramatically by procedure type.
Scale effects: Efficiency gains achieved through higher volume that affect labor, procurement, and overhead.
This manufacturing paradigm enables more sophisticated economic analysis and management approaches, including detailed process mapping, standard time development, and activity-based cost allocation.
The Activity-Based Costing Methodology Applied to IVF
Activity-Based Costing (ABC) provides a structured methodology for analyzing IVF laboratory operations [2]:
Activity mapping: Identify and document all activities performed in each procedure value stream, from initial consultation through embryo transfer or cryopreservation.
Time determination: Calculate both fixed time components (setup, witnessing, cleanup, documentation) and variable time components (per egg or embryo) required by each staff role.
Direct cost calculation: Compute direct costs (labor = time × fully burdened rate, plus supplies) for each activity, incorporating all employee costs including recruitment, training, and professional development.
Indirect cost allocation: Distribute indirect costs (overhead, rent, utilities, gases, liquid nitrogen) and fixed costs (equipment and facility capital charges) based on the relative usage of personnel, equipment, and floor space.
Quality cost assessment: Determine the cost of quality, or rework costs, based on procedure-specific error rates that lead to patient compensation with replacement cycles.
Operational variability analysis: Model how true economic costs vary under different scenarios, such as staff compensation, eggs retrieved, procedure mix, lab scale, and procurement discounts.
This methodology produces a multidimensional cost model that reveals insights impossible to obtain through traditional accounting, demonstrating how costs vary across procedures and within procedures based on biological factors like egg count.
As fertility treatments become increasingly mainstream medical services, the need for sophisticated economic analysis has never been greater. ABC offers a solution, providing a methodological framework that transforms how healthcare providers understand their operations, assess technologies, set prices, and plan for growth [3]. In this work, we demonstrate ABC’s application through detailed examples from IVF laboratory operations.
It is critical to note that our exercise is not meant to provide universal cost benchmarks. Rather, we demonstrate how individual facilities can establish their own ABC framework to properly assess baseline costs, establish realistic target costs, understand the variation in their costs within procedures and across procedures, validate consistent pricing, and determine the impact of operational changes.
A remotely controlled, fully automated ICSI system was developed from off-the-shelf components including an inverted microscope (IX 81; Olympus, USA), a heated stage (Tokai Hit, Japan), a non-contact laser (Lykos DTS, USA), a piezo actuator (PiezoXpert; Eppendorf, Germany), a motorized stage (H117; Prior Scientific Instruments, UK), a motorized objective turret (U-D6BDREMC; Olympus), oil and air microinjectors (CellTram Oil, Eppendorf; Narishighe, Japan) and cameras (IMX477; Arducam, Hong Kong) to allow complete digital supervision (Figure 1). The system implements the principles of laser sperm immobilization (Ebner et al., 2001), laser-assisted ICSI (Abdelmassih et al., 2002; Verza et al., 2013) and piezo-ICSI (Zander-Fox et al., 2021). The previously developed ‘SiD’ AI (Mendizabal-Ruiz et al., 2022) was employed to select and track sperm cells. Additional custom-built software was used to segment cell types such as oocytes and spermatozoa, and microtools. Software was developed to automate key steps, including laser immobilization, sperm loading into the microneedle, oocyte localization and optical segmentation, guided sperm injection, and tracking with focus adjustment on gametes and microtools. Segmentation, in this context, refers to the process of labelling pixels that constitute a specific object, such as a cell. Here, the AI identifies pixels belonging to the spermatozoon and classifies them into ‘head and neck’ or ‘midpiece and tail’ based on their morphological structures. The system was engineered at a dedicated research and development facility (Conceivable Life Sciences, Guadalajara, Mexico).
Evidence Generation from Multi-Clinic Analysis
Site selection and time studiesOur analysis synthesized data from five anonymous IVF clinics across different regions in the USA (see Table 1), representing a range of operational scales (600–2200 annual egg retrieval cycles, underpinning a total of 1100 to 4070 annual ART cycles). Throughout the manuscript, we will refer to center sized by their annual egg retrieval count, since egg retrievals are the primary drivers of lab workload, including any subsequent frozen cycles. The selection of these facilities was based on their willingness to participate under anonymity, and their representation of different geographic regions and operational scales. All centers utilized a skill mix of junior and senior staff, used continuous scheduling rather than batch-mode operations, and did not utilize electronic witnessing.
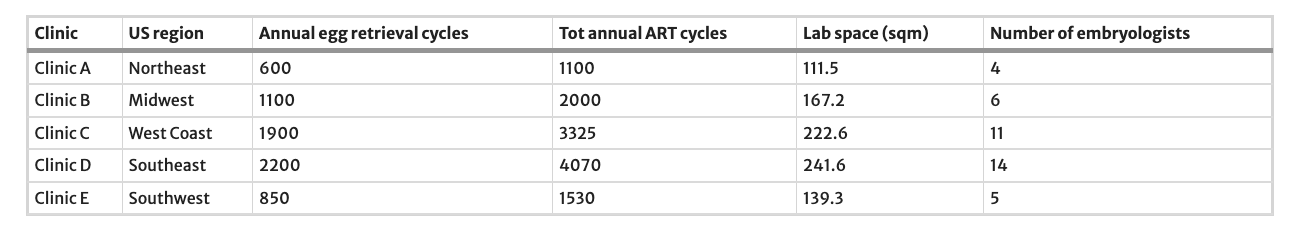
Table 1 Characteristics of the US-based IVF centers evaluated in the analysis. Annual Egg Retrieval Cycles are defined as completed oocyte retrievals per year. Total Annual ART Cycles include all ART procedures per year (egg retrievals plus frozen embryo transfers). The laboratory space calculation encompasses embryology, andrology, cryostorage, lab offices, preparation areas, and all spaces required to support the specified number of lab personnel and work volumes. The capacities shown represent actual operational levels during the study period, not maximum theoretical capacity
Time studies were conducted by the lab director or supervisor at each location between 2022 and 2023, with over 500 procedures observed by senior embryologists across all procedure types at all five clinics, including all preparation, documentation, manual double witnessing, and clean-up activities. Taking the approach used by Veiga and colleagues [4], we calculated the 95th centile (p95) of the process times from all clinics to ensure times reflect typical performance across varying experience levels. We also compared these results to the standards presented in that and other work [4,5,6,7] to ensure the estimates were reasonable and consistent with published data. Facilities implementing ABC should substitute their own time measurements, as absolute values vary with operational factors (staff skill blend, electronic witnessing, automation, stimulation protocols affecting the number of eggs retrieved, among others).
The time studies results revealed significant variations in time requirements across procedures, as reported in Tables 2 and 3.
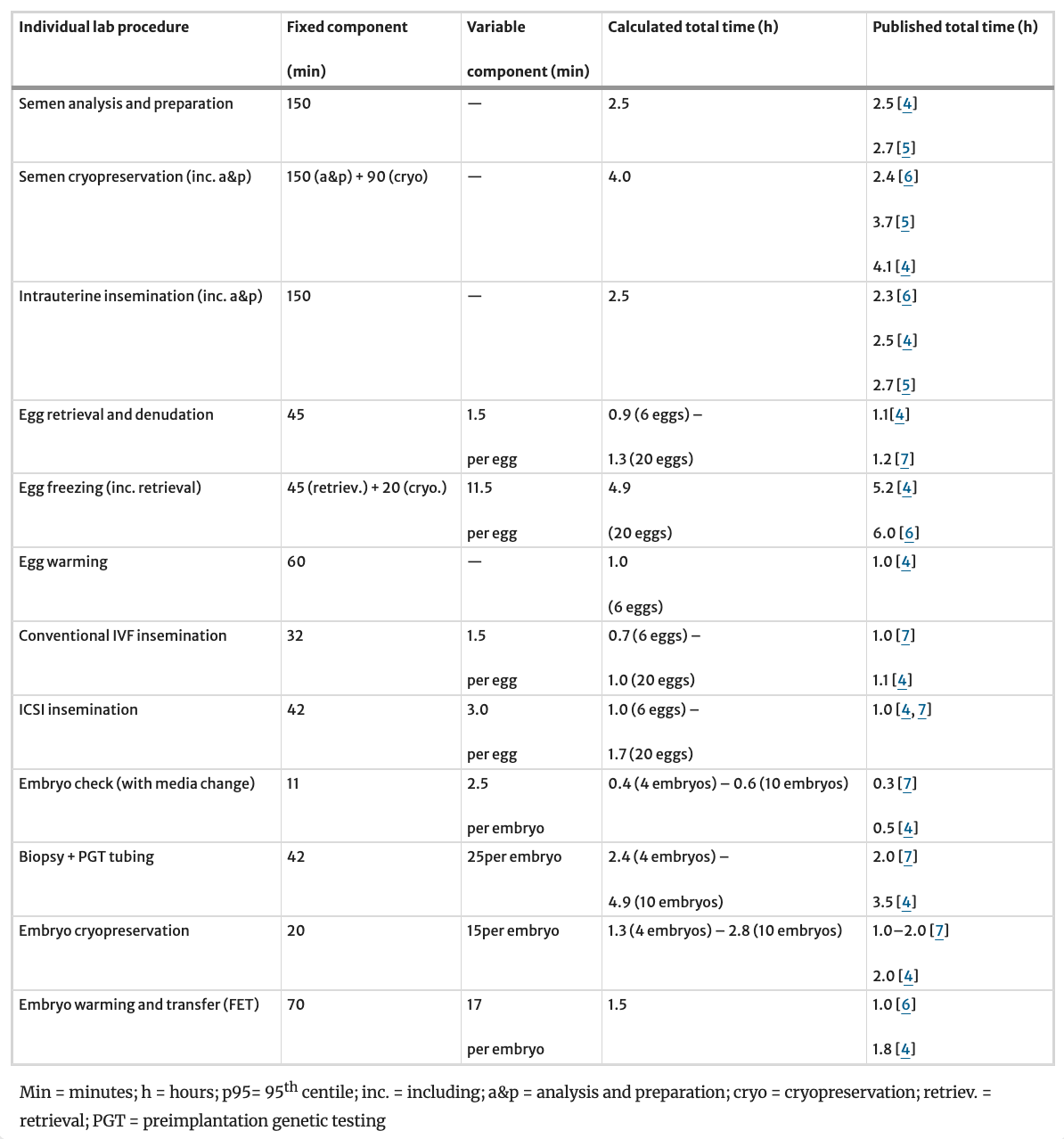
Table 2 Result summary of timed studies at the five participating centers reporting on multiple key laboratory processes. Each reported procedure timing features both a fixed and a variable time component which are combined together to calculate the procedure’s total time. Calculation used the 95th centile for the distribution of each time measurement series to ensure a robust and conservative time estimate. Importantly, timings capture the complete labor cost component of each procedure, including direct procedural time, ancillary administrative tasks, witnessing, setup, cleanup, and necessary idle time. For comparison, timings presented in recent literature for the same procedures are also reported
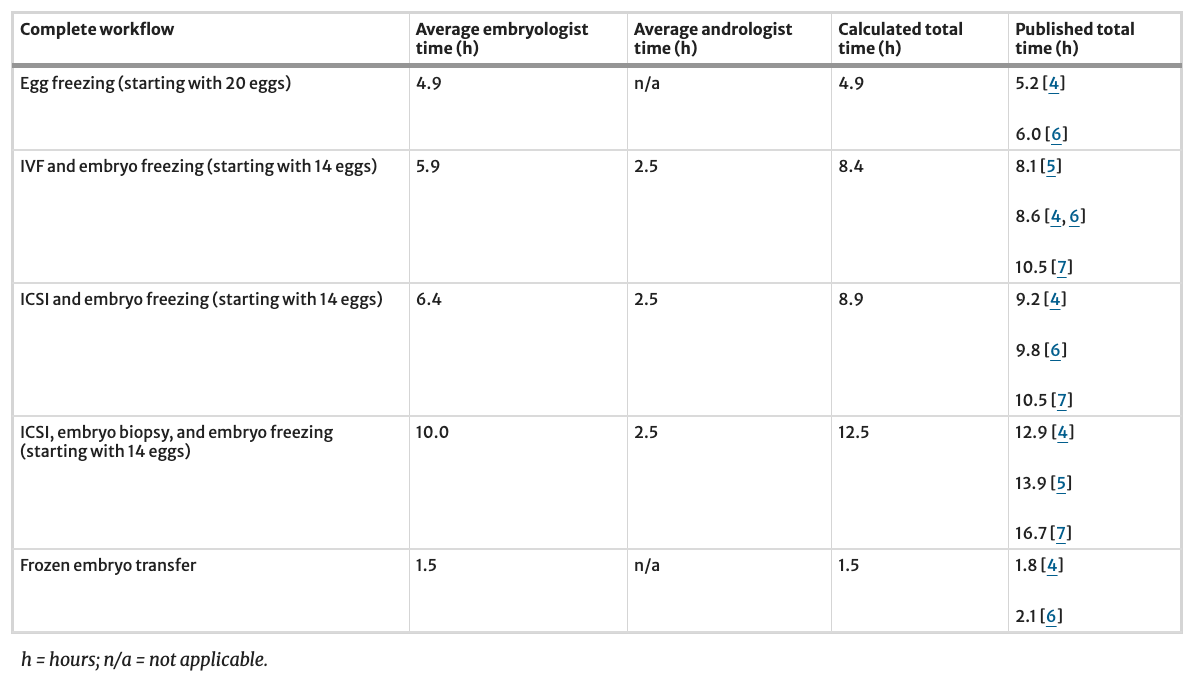
Table 3 Calculated timings for complete laboratory workflows (stringing multiple procedures) offering a breakdown between embryologist and andrologist time contributions. Times represent total laboratory commitment including preparation, procedure execution, monitoring, quality checks, and documentation across the full workflow, not solely hands-on manipulation time. “Complete workflows” include all laboratory activities from sperm/egg processing through cryopreservation, encompassing preparation, insemination, culture monitoring, quality control, and documentation. The table further offers a comparison of our findings against recent published literature
Supply, Equipment, and Capital Charges Data Collection and Validation
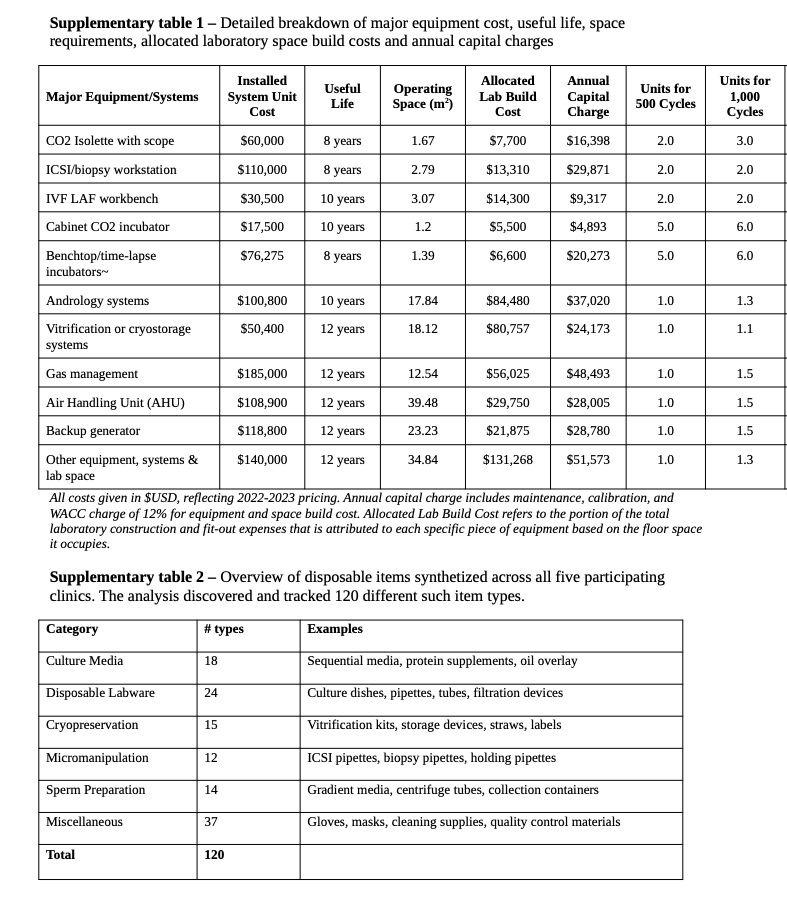
We synthesized supplies and equipment used and their unit prices across all five participating clinics to account for scale, brands, usage per procedure, and prices. Costs were validated against manufacturer catalog, purchasing databases and against published prices online to provide a conservative baseline for a 2000-egg retrieval cycle lab, with price adjustments made based on scale. All costs are reported in US dollars and reflect 2022–2023 pricing. Further details on supply and equipment components are available in Supplementary Tables 1 and 2.
Capital charges included equipment depreciation calculated using a useful life estimate for major equipment (see Supplementary Table 1), annual maintenance contracts typically representing 12–15% of equipment value, and a weighted average cost of capital (WACC) of 12% applied to both equipment and facility build-out costs. Equipment utilization varies significantly by procedure type, with complex procedures requiring specialized instrumentation for extended periods, which was accounted for in our model. All overhead costs were included in our analysis as they represent real economic costs that must be covered for facility operations, regardless of payment source or accounting treatment. The ABC methodology allocated these costs based on actual resource consumption patterns rather than arbitrary allocation methods.
Labor Economies of Scale: A Queueing Theory Perspective
Labor costs (in US dollars) incorporated fully burdened hourly rates (including gross salary, benefits, payroll, employer taxation, training, travel, recruitment, and replacement): embryologists (blend of senior and junior) at $108/h and andrologists/lab technicians at $66/h, with lab director compensation allocated across all procedures. These labor costs are representative of the actual skill blend at our participating centers, but we anticipate a wide national variability.
Our ABC model applied the generally accepted principle that networks of parallel process staff resources exhibit significant economies of scale when handling increasing workloads due to fundamental queueing theory principles [8]. Healthcare applications demonstrate these principles, where consolidated service centers achieve better resource utilization than distributed facilities [9]. The Halfin-Whitt heavy-traffic regime formalized these economies of scale, showing that optimal staffing levels grow sub linearly with demand while maintaining service quality [10].
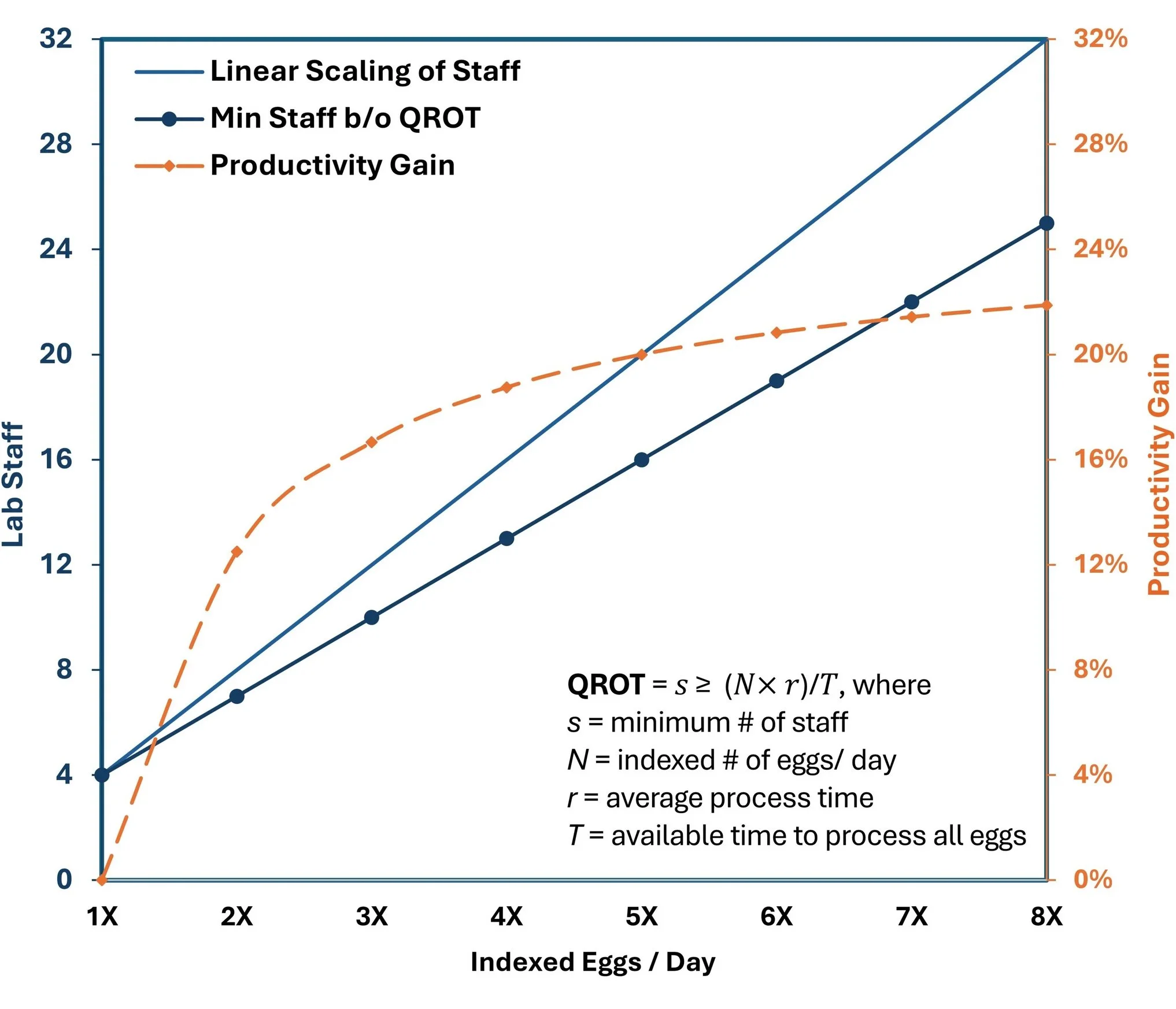
To simplify the application of queuing network theory and make staff utilization calculations more accessible for practical IVF laboratory management, we employed the Queuing Rule of Thumb (QROT) [11]. QROT is a simplified approximation derived from queuing theory that provides a conservative estimate of resource requirements by assuming all demand arrives simultaneously at the beginning of service. The formula (N = s × r/T) relates the number of customers (N), servers (s), service time (r), and total time available to process customers (T). While less precise than full stochastic queuing formulas, QROT offers practitioners a straightforward tool for capacity planning without requiring complex computations or in-depth knowledge of queuing theory.
In our fertility center context, QROT allowed us to model how staff productivity improves with scale by treating eggs retrieved as “customers” and laboratory staff as “servers” in a queuing system. The QROT analysis showed that as clinic volume increases from a modeled 500 to 4000 annual egg retrieval cycles, the productivity gain from increased pooling of resources and processing times of eggs retrieved improves staff utilization rates from 72.5% to 80%, revealing the near-linear relationship between volume and efficiency. This occurs because larger centers can better absorb demand variability, minimizing overall idle time. The model thus quantifies why larger fertility centers achieve larger productivity gains (see Fig. 1). This theoretical foundation directly supports the 30% labor efficiency improvement observed between 500 and 2000 egg retrieval cycle operations in our empirical analysis.
Figure 1 Queuing Rule of Thumb (QROT) analysis of staff productivity gain. Centers performing a higher number of egg collections per day use proportionally less staff time per procedure
ABC in Practice: Modeling Cost Structures and Variations
The ABC methodology’s value lies not in revealing universal cost relationships, but in enabling facilities to model how these relationships change under different operational scenarios. The ability to derive context-dependent insights, rather than the absolute values presented in the example applications below, represents the methodology’s primary contribution.
In our example applications, we focus in particular on six representative laboratory processes:
Egg freezing
Egg warming, ICSI, and embryo culture and freezing
Conventional IVF, and embryo culture and freezing
ICSI, and embryo culture and freezing
ICSI, embryo culture, biopsy, and freezing
Frozen embryo transfer
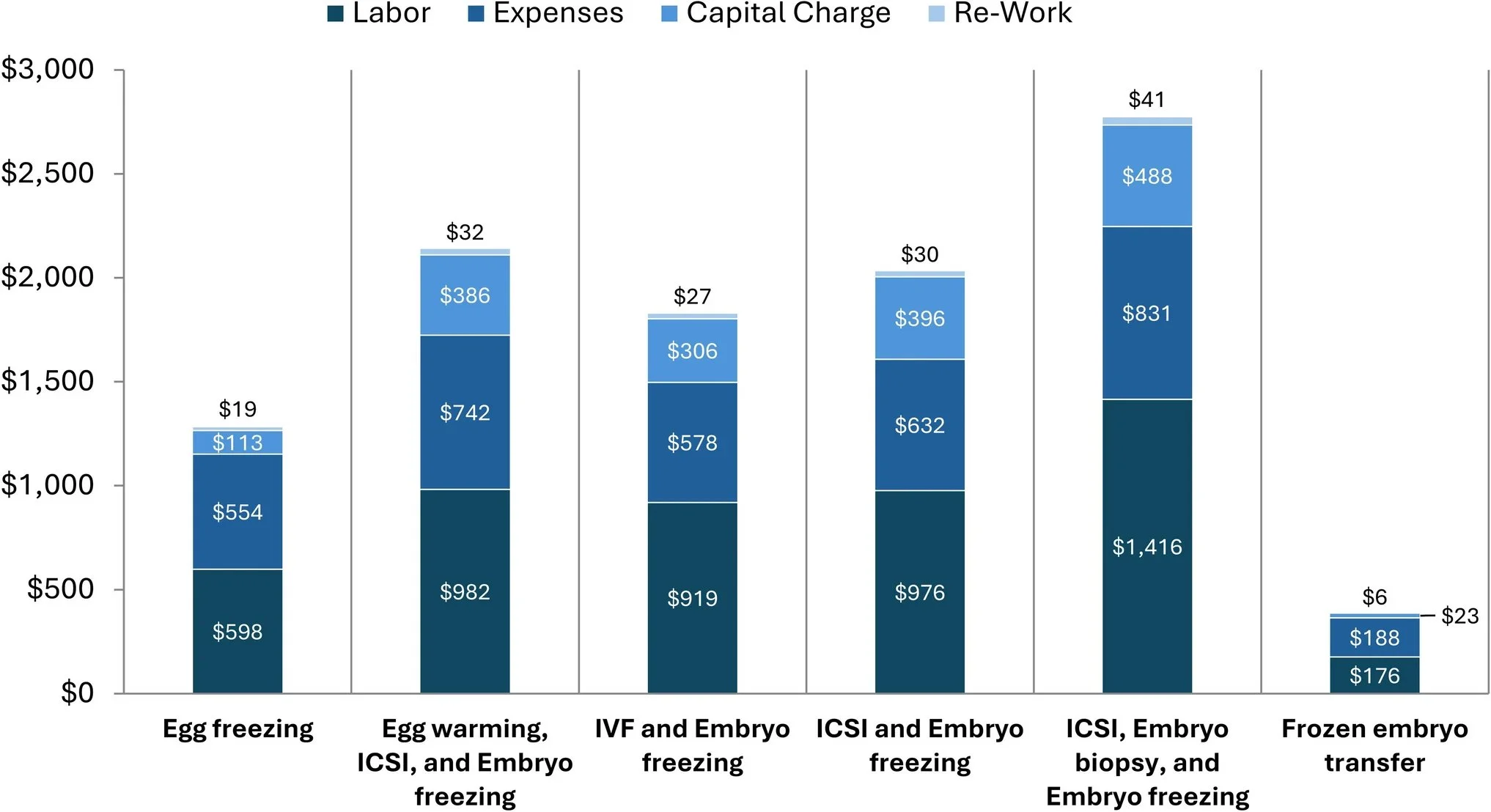
The analysis reveals dramatic variation in procedure costs that traditional accounting methods might miss. While simplistic accounting relying on procedure counts might calculate an average labor cost of approximately $1126 per procedure, our ABC analysis (using the hourly rates and time study methodology detailed above) shows actual labor costs ranging from $598 to $1416 depending on the specific procedure. Similarly, when indirect expenses are fully allocated, expenses per procedure average $686 but vary considerably by procedure type. Capital charges, often overlooked in operational decisions, average $465 per procedure but show significant procedure-dependent variation. Overall, total costs per procedure vary substantially, with factors like ICSI and biopsy increasing labor, equipment, and supply costs. Figure 2 reveals the dramatic variation in procedure costs across different types of IVF services in a center performing 2000 egg retrieval cycles per year, with an additional breakdown by cost component.
Figure 2 Average costs by procedure type at a center performing 2000 yearly egg retrieval cycles
Examples of Cost Elasticity Calculations with Biological Inputs
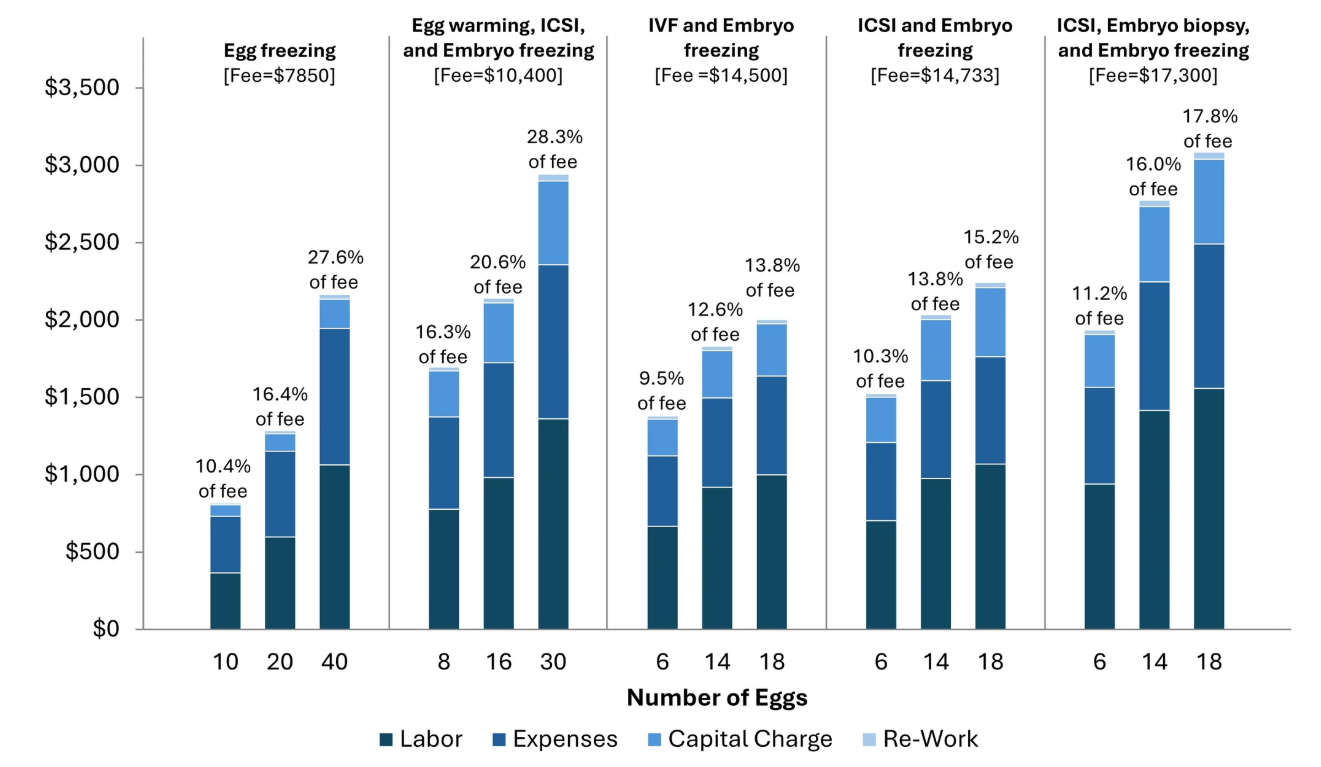
Even more revealing is how costs vary dramatically within the same procedure based on biological factors. Figure 3 illustrates how procedure costs increase with egg count and decrease with lab scale, demonstrating the concept of “cost elasticity” (CE), the percentage change in cost relative to the percentage change in input volume calculated as (% change in cost)/(% change in input volume). For example:
For egg freezing, costs rise by 165% when the number of eggs increases by 300% (from 10 to 40 eggs) resulting in a cost elasticity of 55%. Specifically, the cost increases from $642 (for 10 eggs) to $1698 (for 40 eggs), a difference of $1056. This represents a 165% increase in cost (1698 − 642) ÷ 642 × 100.
For ICSI with embryo freezing, costs increase by 47% when egg volume increases by 200% (from 6 to 18 eggs), giving a cost elasticity of 24%.
For ICSI cycles including embryo biopsy, costs increase by 59% when egg volume increases by 200%, giving a cost elasticity of 30%.
Figure 3 Cost variation by egg count for different procedures at a center offering 2000 egg retrieval cycles/year. A typical center charges a fixed fee while costs vary with the number of eggs retrieved. For each procedure, we present the average fees charged across participating centers and calculate what percentage of this fee is used to cover for true lab costs based on biological input
Scale Economics
Our empirical analysis encompasses facilities performing 600–2200 annual egg retrieval cycles. Projections for a 4000-egg retrieval cycle facility represent theoretical extensions based on QROT and established healthcare consolidation patterns, not direct observations. These projections likely underestimate actual economies of scale, as larger facilities typically implement additional efficiencies (electronic systems, specialized support staff, automation) not captured in our conservative model. In our example ABC application, we present these as directional indicators of scale benefits rather than precise predictions. Our analysis revealed several key insights:
Labor efficiency increases with scale but plateaus: Embryologist productivity (measured in procedures per FTE) increases approximately 30% when moving from 500 to 2000 yearly egg retrieval cycles, but gains diminish beyond that point.
Supply costs decrease continuously with scale: Volume discounts accumulate as labs grow, with the largest labs achieving 10–15% lower supply costs than mid-sized operations. However, network-affiliated smaller labs may achieve procurement advantages similar to larger independent facilities so readers should be cautious about applying this conclusion widely without performing ABC assessment in their specific use case
Capital utilization improves dramatically with scale: Equipment utilization rates in 4000-egg retrieval cycle labs are 1.8 to 2.2 times higher than in 500-cycle labs, creating substantial cost advantages.
Space efficiency follows a similar pattern: Space per egg retrieval cycle decreases by approximately 40% when moving from a 500-cycle to a 2000-cycle lab.
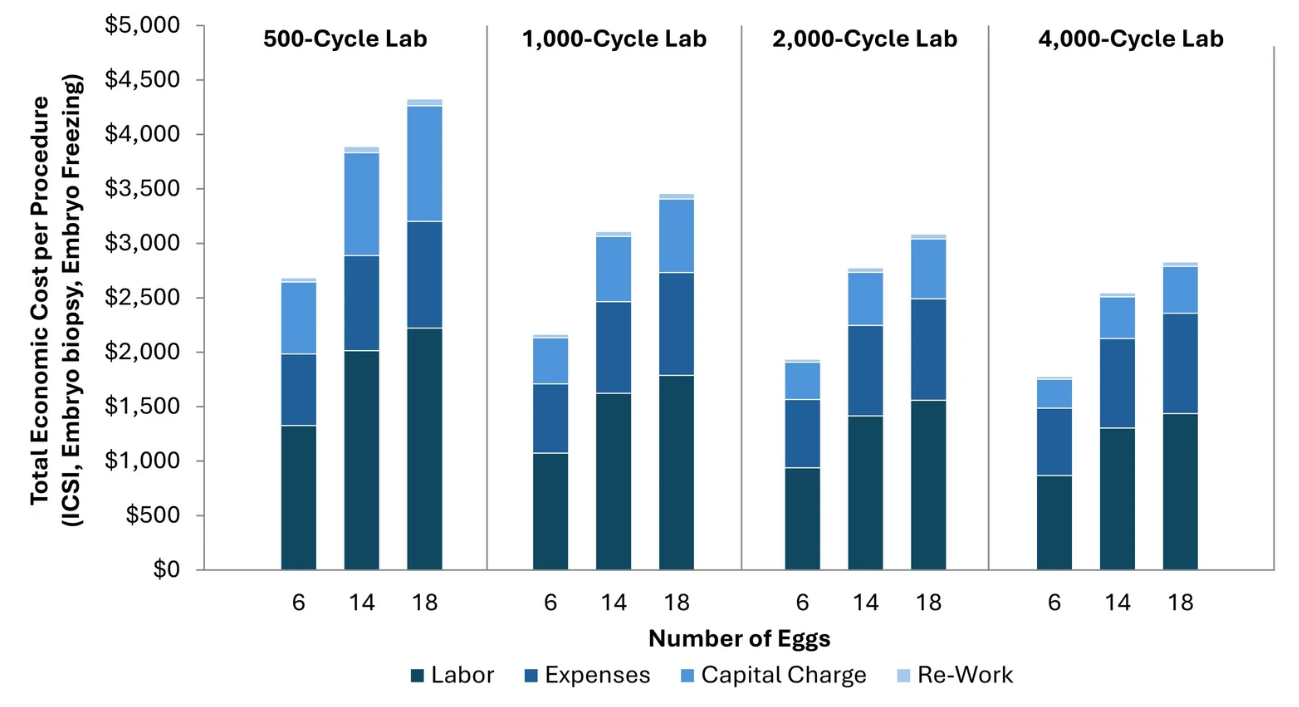
Table 4 provides an overview of cost components and their calculated variation with lab scale. When combined, these factors translate to overall cost advantages of 40% for large labs compared to small labs, providing economic rationale for the consolidation trend in the fertility sector. Figure 4 extends on this analysis, showing the impact of biological inputs on the same trend by focusing on a single, complex lab procedure (ICSI with embryo culture, biopsy, and freezing).
Figure 4 A comparative overview for the total economic cost of ICSI and embryo biopsy plus embryo cryostorage between IVF centers of different sizes (500 to 4000 egg retrieval cycles per year), based on number of eggs treated. Larger centers typically achieve greater cost-efficiency
Strategic applications of the ABC methodology
Our analysis focuses on understanding true underlying economic costs, which exist regardless of billing mechanisms, reimbursement structures, or CPT code constraints. This cost transparency empowers clinics to make informed decisions about insurance participation, contract negotiations, and service delivery strategies.
Most IVF clinics use flat-rate pricing regardless of the expected number of eggs retrieved or embryos created. ABC analysis reveals that the actual laboratory costs vary significantly based on case complexity and egg number. This cost variation presents an opportunity for clinics to explore alternative pricing models that better reflect resource utilization. With transparency into true costs, clinics can develop more sophisticated pricing models that optimize financial sustainability while ensuring all patients receive the same high-quality care regardless of their clinical characteristics. Such models could potentially include volume-based laboratory fees or tiered pricing structures that maintain equitable access to treatment. By understanding true costs, clinics can also make more informed decisions about participation in insurance networks, guarantees, and financing programs. Importantly, ABC is intended to inform operational efficiency, not clinical treatment. Used correctly, ABC supports quality of care and patient outcomes as the primary considerations in fertility services, as further discussed below.
Lab Capacity Planning and Workload Management
The ABC methodology demonstrates that workload variation is not simply a function of egg retrieval cycle count but depends on procedure type and egg/embryo volume. Traditional scheduling approaches that focus only on the number of retrievals per day can lead to significant workload imbalances, creating stress and quality risks on high-volume days and inefficient resource utilization on others [12].
With detailed time standards for each procedure and awareness of how time requirements scale with egg/embryo count, labs can develop sophisticated scheduling algorithms that balance workload across days and weeks. This improves staff satisfaction, reduces error rates, and maximizes equipment utilization.
Technology Assessment and Capital Investment
ABC provides a rigorous framework for evaluating new technologies and process improvements. By understanding the detailed cost components of each procedure, clinics can calculate the true return on investment for equipment upgrades, automation systems, and process changes, while evaluating cost-effectiveness. In a theoretical example, the ABC analysis might reveal that a new time-lapse imaging system that does not to affect clinical outcomes increases capital costs but reduces labor requirements by eliminating manual observations. This tradeoff might be economically advantageous in high-labor-cost regions but disadvantageous in others. Similarly, automation that reduces variable time per egg becomes increasingly valuable as egg counts rise, potentially justifying different technology choices for clinics with different patient demographics.
Scaling Strategies and Consolidation Economics
The ABC analysis quantifies the economic benefits of scale, showing how larger labs spread fixed costs across more procedures. This provides a data-driven foundation for consolidation strategies, network planning, and hub-and-spoke models where large central IVF facilities performing all laboratory procedures (hubs) collaborate with a distributed network of clinics (spokes) where patient evaluation and ovarian stimulation take place [13].
By understanding the cost elasticity of different procedures, organizations can determine which services should be centralized in high-volume hubs and which can be efficiently delivered in smaller satellite locations. The analysis also reveals which costs decrease most significantly with scale (typically capital and facility costs) and which remain relatively constant (typically direct labor and supplies), informing strategic decisions about investment priorities.
Business Model Innovation
By revealing the true economics of different procedures and patient profiles, ABC analysis enables innovation in service delivery models. For example, the analysis might quantify the resources needed to perform egg freezing for patients with higher egg counts, suggesting an opportunity for clinics to offer tiered pricing that makes the service more accessible to women with normal or high ovarian reserve, underpinning subscription models to distribute costs across a patient’s fertility journey, and creating evidence-based financing that aligns with procedure economics.
Implementation Challenges and Considerations
Implementing ABC in IVF operations faces several challenges:
Data collection complexity: Gathering accurate time standards for each activity requires careful observation and documentation. Manual time studies, video analysis, or electronic tracking systems may be needed.
Staff engagement: Embryologists and andrologists may initially resist time tracking, fearing it will lead to unrealistic productivity targets. Clear communication about the purpose and benefits of the analysis is essential.
Activity definition granularity: Determining the right level of detail for activity mapping requires balancing precision with practicality. Too much detail becomes unwieldy, while too little sacrifices insight.
Cost allocation methods: Allocating indirect costs requires thoughtful methodology development. Different allocation bases (time, space, equipment usage) yield different results, and the approach should reflect operational realities.
Continuous updating: As technologies, protocols, and costs evolve, the ABC model must be regularly updated to maintain accuracy.
Ethical implementation
It is crucial to emphasize that ABC analysis is a financial management tool, not a clinical decision-making framework. ABC data should inform operational efficiency and pricing strategy at the institutional level, never individual patient care decisions. Clinics must ensure that cost awareness does not create implicit bias in treatment protocols or staff behavior.
Despite these challenges, the potential benefits make implementation worthwhile for most IVF facilities, particularly those seeking to scale operations or understand their true economics.
Limitations
Our analysis has several constraints that readers should consider. First, time estimates reflect conservative operational conditions (manual witnessing, no automation) that may not represent all facilities. Second, projections beyond 2200 egg retrieval cycles/year, while grounded in established theory, lack empirical validation. These theoretical extensions demonstrate how the ABC framework can model scale effects but should not be interpreted as empirically validated benchmarks. Third, cost structures reflect US market conditions and may not translate directly to other healthcare systems. These limitations do not diminish the methodology’s value but rather emphasize that each facility must develop context-specific models using the ABC framework we exemplify.
Our current assessment focuses specifically on laboratory operations, which represent a substantial but incomplete picture of total IVF costs. Clinical operations typically represent an additional 1.5 times the laboratory costs we analyze here. The ABC methodology and theoretical frameworks demonstrated for laboratory operations can be adapted and extended to clinical activities, though the complexity of clinical decision-making and patient variability would require additional methodological considerations beyond the scope of this analysis.
Conclusions
transparency and economic sustainabilityIVF demand continues to grow as awareness increases, social norms evolve, and reproductive challenges persist, suggesting an expansion of the service offer will be necessary [14]. However, high costs limit access, and opacity in pricing creates market inefficiencies. The SART indicates that the average livebirth rate per retrieval cycle was 35.3% in 2022, implying the average US patient requires 2.8 retrieval cycles for a baby, with significant variability due to age (https://www.sartcorsonline.com/rptcsr_publicmultyear.aspx, accessed 15th July 2025). In our own assessment, the laboratory cost of an ICSI cycle with embryo biopsy and cryopreservation is $2775 per cycle—and so the average of 2.8 cycles to baby attracts a total lab cost of $7770. The industry remains highly fragmented, with most fertility centers maintaining their own small embryology labs.
ABC reveals the true cost drivers of IVF procedures, documenting significant cost elasticity and scale economies that provide economic rationale for sector consolidation. These insights enable more sophisticated pricing models that could expand patient access while maintaining financial sustainability.
The methodology presented here offers a template for analyzing other specialized healthcare services that combine variable biological inputs, specialized capital, and skilled labor. Most importantly, by providing transparency into true operational costs, ABC analysis transforms complex operational data into actionable strategic insights.
Future research should extend this methodology to clinical operations and explore how ABC-informed pricing and operational strategies affect patient outcomes and access to care. The ultimate goal is not simply cost reduction but the optimization of value creation in fertility services.
Data Availability
The data underscoring this work is available from the corresponding author upon reasonable request.
-
Hariton E, et al. Meeting the demand for fertility services: the present and future of reproductive endocrinology and infertility in the United States. Fertil Steril. 2023;120:755-766.
Cassettari L, et al. IVF cycle cost estimation using activity-based costing and Monte Carlo simulation. Health Care Manag Sci. 2016;19:20-30.
Keel G, et al. Time-driven activity-based costing in health care: a systematic review of the literature. Health Policy. 2017;121:755-763.
Veiga E, et al. Recalculating the staff required to run a modern assisted reproductive technology laboratory. Hum Reprod. 2022;37:1774-1785.
Olmed Illueca C, et al. P-161: Is enough the staff in your lab? Hum Reprod. 2021;36(Suppl 1):deab130.160.
Lee YS, et al. Guidelines for best practice for staffing of ART laboratories and professional development of IVF scientists. Fertil Reprod. 2023;31:163-175.
Alikani M, et al. Comprehensive evaluation of contemporary assisted reproduction technology laboratory operations to determine staffing levels that promote patient safety and quality care. Fertil Steril. 2014;102:1350-1356.
Mulligan JG. The economies of massed reserves. Am Econ Rev. 1983;73:725-734.
Green L. Queueing analysis in health care. In: Hall R, ed. Patient Flow. Int Ser Oper Res Manag Sci. 2013;206.
van Leeuwaarden JS, et al. Economies-of-scale in many-server queueing systems: tutorial and partial review of the QED Halfin-Whitt heavy-traffic regime. SIAM Rev. 2019;61:403-440.
Teknomo K. Queuing rule of thumb based on M/M/s queuing theory with applications in construction management. Civ Eng Dimens. 2012;14:139-146.
Basar M, Duzcu T. The burnout: a silent saboteur in in vitro fertilization laboratories. J Assist Reprod Genet. 2025.
Berger DS, et al. Remote IVF: a clinical and laboratory guide to performing remote oocyte retrievals. J Assist Reprod Genet. 2023;40:2081-2089.
Kushnir VA, et al. The future of IVF: the new normal in human reproduction. Reprod Sci. 2022;29:849-856.
-
Authors and Affiliations
Conceivable Life Sciences, 160 Mercer St., New York, NY, 10012, USA
Alejandro Chavez-Badiola, Giuseppe Silvestri & Alan Murray
MonCap, Boulder, CO, USA
Steve Rooks
Contributions
Alejandro Chavez Badiola: conceptualization, subpervision, writing—original draft, writing—review and editing. Steve Rooks: conceptualization, data curation, formal analysis, methods, resources, writing—review and editing. Giuseppe Silvestri: visualization, writing—review and editing. Alan Murray: conceptualization, project administration, resources, writing—review and editing.
Corresponding author
Correspondence to Alejandro Chavez-Badiola.
-
Ethics approval and consent to participate
Not applicable.
Competing interests
Financial interests: ACB and AM are company officers at Conceivable Life Sciences. ACB, GS, and AM are stockholders and patent inventors at Conceivable Life Sciences. ACB is also a stockholder of IVF 2.0. AM is also a stockholder of TMRW Life Sciences. SR has received consultation fees from Conceivable Life Sciences. GS received consultation fees from IVF 2.0 within the 36 months prior to this manuscript, unrelated to the present work.
Non-financial interests: none.
Supplementary Information:
Key Message
Applying Activity-Based Costing (ABC) to IVF laboratories reveals that fertility labs operate as highly specialized manufacturing systems—where scale, workflow design, labor allocation, and technology investment directly shape cost, efficiency, and clinical capacity. By making cost structures transparent and modeling biological and operational variability, ABC provides a strategic framework for smarter capacity planning, capital deployment, and sustainable growth—while maintaining ethical and clinical oversight.